View in other NatureServe Network Field Guides
NatureServe
Montana
Utah
Wyoming
Idaho
Wisconsin
British Columbia
South Carolina
Yukon
California
New York
Bitterroot Milkvetch - Astragalus scaphoides
State Rank Reason (see State Rank above)
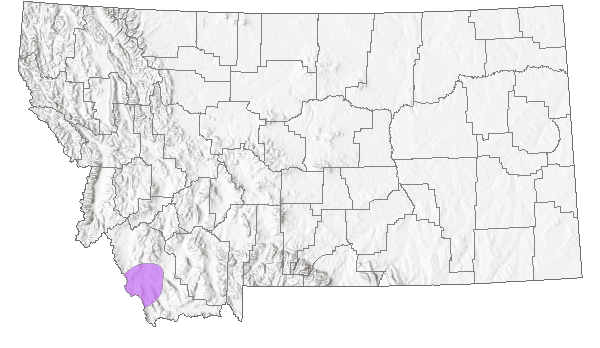
Bitterroot milkvetch occurs only in Lemhi County, Idaho and Beaverhead County, Montana. In Montana, the documented occurrences are confined to an area from the Grasshopper Creek drainage south to the Tendoy Mountains. The total number of individual plants has been estimated in the tens of thousands, but occupied habitat is likely less than 700 acres.
General Description
Bitterroot milkvetch is a stout, herbaceous perennial with several erect stems 2-6 dm in height arising from a branched rootcrown. The pinnately compound leaves (10-25 cm long) have 15-21 narrowly elliptic leaflets, and the foliage is glabrous to sparsely hairy. Crowded clusters of 15-30 spreading yellowing flowers are borne in the axils of upper leaves, becoming less dense as the plant matures. Corollas are about 20 mm long and have a reflexed upper petal and a blackish-hairy calyx (8-10 mm long). Glabrous, green to reddish, oblong pods (15-20 mm long by 4-6 mm wide) are 2-chambered in cross-section, and are borne on a stalk of approximately equal length that spreads out and then arches up, holding the fruit nearly erect.
Phenology
Flowering occurs in late May-early June, with fruit maturing in July.
Diagnostic Characteristics
Astragalus atropubescens is similar, but has white flowers and smaller diameter (2-4 mm) fruits borne on sharply erect (immediately ascending) stalks. Astragalus canadensis also differs in having sharply erect fruit stalks. Astragalus terminalis has white flowers and unstalked fruits.
Species Range
Montana Range
Range Descriptions
 Native
Native
Range Comments
Beaverhead County, Montana, and Lemhi County, Idaho.
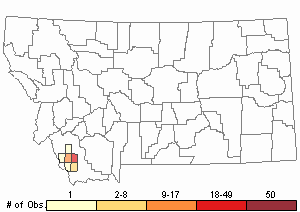
Observations in Montana Natural Heritage Program Database
Number of Observations: 86
(Click on the following maps and charts to see full sized version)
Map Help and Descriptions
Relative Density
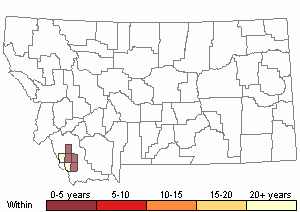
Recency
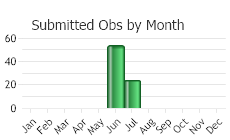
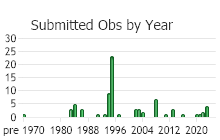
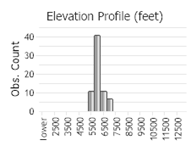 (Observations spanning multiple months or years are excluded from time charts)
(Observations spanning multiple months or years are excluded from time charts)
Habitat
Bitterroot milkvetch occurs in sagebrush grassland, generally with a dense cover of sagebrush on silty soils with a moderate to high content of coarse material derived from limestone, basalt or diabase (Lesica 1984). Populations are often found along drainages in the ecotonal area between rocky, steep upper slopes and nearly level benches along drainageways, and seem to be most frequent on warmer, south- and southwest-facing slopes. The habitat usually has a high percentage of bare ground, and shrubs other than sagebrush sometimes dominate. Frequent associates include Artemisia tridentata, Chrysothamnus nauseosus, Artemisia nova, Agropyron spicatum, Agropyron smithii, Stipa comata, Oryzopsis hymenoides, Opuntia polyacantha, Gutierrezia sarothrae, Phlox hoodii and Artemisia frigida.
National Vegetation Classification System Groups Associated with this Species
Shrubland
Foothills - Montane Shrubland
Sagebrush Shrubland
Ecology
The number of above-ground stems is highly variable among years, making accurate population assessments difficult (Lesica 1995).
POLLINATORS The following animal species have been reported as pollinators of this plant species or its genus where their geographic ranges overlap:
Bombus vagans,
Bombus appositus,
Bombus auricomus,
Bombus bifarius,
Bombus borealis,
Bombus centralis,
Bombus fervidus,
Bombus flavifrons,
Bombus huntii,
Bombus mixtus,
Bombus nevadensis,
Bombus rufocinctus,
Bombus ternarius,
Bombus terricola,
Bombus occidentalis,
Bombus pensylvanicus,
Bombus griseocollis, and
Bombus insularis (Macior 1974, Thorp et al. 1983, Mayer et al. 2000, Colla and Dumesh 2010, Wilson et al. 2010, Koch et al. 2012, Miller-Struttmann and Galen 2014, Williams et al. 2014).
Management
The tendency of this species to occur on lower slopes above drainages makes it particularly vulnerable to road construction (Lesica 1984), which has likely impacted some populations.
Herbivory by insects and mammals may limit the reproductive ability of this species (Lesica 1984, 1995). However, three sample populations remained stable or increased in size in spite of significant seed predation (Lesica 1995). Plants are most vulnerable to grazing as they mature, between May 15 and July 15. Limiting livestock grazing during this period minimizes soil compaction and damage to reproductive plants (Lesica 1984). Rest-rotation grazing regimes may allow enough recruitment to maintain stable populations, while chronic severe grazing could reduce adult vigor and lead to population decline (Lesica 1995).
Stewardship Responsibility
Threats or Limiting Factors
STATE THREAT SCORE REASON
Reported threats to Montana’s populations of Bitterroot Milkvetch are currently assigned as unknown (MTNHP Threat Assessment 2021). Spotted Knapweed (Centaurea stoebe) is found in the vicinity of some populations, but reports indicate that severity of impacts is unknown. Information about the severity of impacts to populations due to noxious weeds or other threats is needed.
References
- Literature Cited AboveLegend:
 View Online Publication
View Online Publication Colla, S.R. and S. Dumesh. 2010. The bumble bees of southern Ontario: notes on natural history and distribution. Journal of the Entomological Society of Ontario 141:39-68.
Colla, S.R. and S. Dumesh. 2010. The bumble bees of southern Ontario: notes on natural history and distribution. Journal of the Entomological Society of Ontario 141:39-68. Koch, J., J. Strange, and P. Williams. 2012. Bumble bees of the western United States. Washington, DC: USDA Forest Service, Pollinator Partnership. 143 p.
Koch, J., J. Strange, and P. Williams. 2012. Bumble bees of the western United States. Washington, DC: USDA Forest Service, Pollinator Partnership. 143 p. Lesica, P., M.T. Lavin, and P.F. Stickney. 2012. Manual of Montana Vascular Plants. Fort Worth, TX: BRIT Press. viii + 771 p.
Lesica, P., M.T. Lavin, and P.F. Stickney. 2012. Manual of Montana Vascular Plants. Fort Worth, TX: BRIT Press. viii + 771 p. Macior, L.M. 1974. Pollination ecology of the Front Range of the Colorado Rocky Mountains. Melanderia 15: 1-59.
Macior, L.M. 1974. Pollination ecology of the Front Range of the Colorado Rocky Mountains. Melanderia 15: 1-59. Mayer, D.F., E.R. Miliczky, B.F. Finnigan, and C.A. Johnson. 2000. The bee fauna (Hymenoptera: Apoidea) of southeastern Washington. Journal of the Entomological Society of British Columbia 97: 25-31.
Mayer, D.F., E.R. Miliczky, B.F. Finnigan, and C.A. Johnson. 2000. The bee fauna (Hymenoptera: Apoidea) of southeastern Washington. Journal of the Entomological Society of British Columbia 97: 25-31. Miller-Struttmann, N.E. and C. Galen. 2014. High-altitude multi-taskers: bumble bee food plant use broadens along an altitudinal productivity gradient. Oecologia 176:1033-1045.
Miller-Struttmann, N.E. and C. Galen. 2014. High-altitude multi-taskers: bumble bee food plant use broadens along an altitudinal productivity gradient. Oecologia 176:1033-1045. MTNHP Threat Assessment. 2021. State Threat Score Assignment and Assessment of Reported Threats from 2006 to 2021 for State-listed Vascular Plants. Botany Program, Montana Natural Heritage Program, Helena, Montana.
MTNHP Threat Assessment. 2021. State Threat Score Assignment and Assessment of Reported Threats from 2006 to 2021 for State-listed Vascular Plants. Botany Program, Montana Natural Heritage Program, Helena, Montana. Thorp, R.W., D.S. Horning, and L.L. Dunning. 1983. Bumble bees and cuckoo bumble bees of California (Hymenoptera: Apidae). Bulletin of the California Insect Survey 23:1-79.
Thorp, R.W., D.S. Horning, and L.L. Dunning. 1983. Bumble bees and cuckoo bumble bees of California (Hymenoptera: Apidae). Bulletin of the California Insect Survey 23:1-79. Williams, P., R. Thorp, L. Richardson, and S. Colla. 2014. Bumble Bees of North America. Princeton, NJ: Princeton University Press. 208 p.
Williams, P., R. Thorp, L. Richardson, and S. Colla. 2014. Bumble Bees of North America. Princeton, NJ: Princeton University Press. 208 p. Wilson, J.S., L.E. Wilson, L.D. Loftis, and T. Griswold. 2010. The montane bee fauna of north central Washington, USA, with floral associations. Western North American Naturalist 70(2): 198-207.
Wilson, J.S., L.E. Wilson, L.D. Loftis, and T. Griswold. 2010. The montane bee fauna of north central Washington, USA, with floral associations. Western North American Naturalist 70(2): 198-207.
- Additional ReferencesLegend:
 View Online Publication
View Online Publication
Do you know of a citation we're missing? Barneby, R.C. 1964. Atlas of North American Astragalus. 2 Vols. New York Botanical Garden, Bronx, New York. 1188 pp.
Barneby, R.C. 1964. Atlas of North American Astragalus. 2 Vols. New York Botanical Garden, Bronx, New York. 1188 pp. DuBois, K.L. 1979. An inventory of the avifauna in the Long Pines of Southeastern Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 113 p.
DuBois, K.L. 1979. An inventory of the avifauna in the Long Pines of Southeastern Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 113 p. Horn, G. 1980. Report on inventory of threatened or endangered, rare, or sensitive plants-Headwaters Resource Area, Butte District, Bureau of Land Management, Montana. Unpublished report. 54 pp.
Horn, G. 1980. Report on inventory of threatened or endangered, rare, or sensitive plants-Headwaters Resource Area, Butte District, Bureau of Land Management, Montana. Unpublished report. 54 pp. Lesica, P. 1984. Report on the Conservation Status of A. scaphoides, a Candidate Threatened Species (Unpublished).
Lesica, P. 1984. Report on the Conservation Status of A. scaphoides, a Candidate Threatened Species (Unpublished). Lesica, P. 1991. Demographic Monitoring of Astragalus Scaphoides At Sheep Corral Gulch, Beaverhead County, Montana: 1991 Progress Report. Unpublished Report to the Bureau of Land Management. Montana Natural Heritage Program, Helena, Mt. 20 Pp.
Lesica, P. 1991. Demographic Monitoring of Astragalus Scaphoides At Sheep Corral Gulch, Beaverhead County, Montana: 1991 Progress Report. Unpublished Report to the Bureau of Land Management. Montana Natural Heritage Program, Helena, Mt. 20 Pp. Lesica, P. 1993. Demographic Monitoring of Astragalus Scaphoides At Two Sites in Montana and Idaho: 1992 Progress Report. Prepared for Bureau of Land Management. Montana Natural Heritage Program, Helena, Mt. 16 Pp.
Lesica, P. 1993. Demographic Monitoring of Astragalus Scaphoides At Two Sites in Montana and Idaho: 1992 Progress Report. Prepared for Bureau of Land Management. Montana Natural Heritage Program, Helena, Mt. 16 Pp. Lesica, P. 1994. Demographic monitoring of Astragalus scaphoides at two sites in Montana and Idaho: final report. Unpublished report for the Bureau of Land Management, Butte District. Montana Natural Heritage Program, Helena, MT.
Lesica, P. 1994. Demographic monitoring of Astragalus scaphoides at two sites in Montana and Idaho: final report. Unpublished report for the Bureau of Land Management, Butte District. Montana Natural Heritage Program, Helena, MT. Lesica, P. 1995. Demography of Astragalus scaphoides and effects of herbivory on population growth. Great Basin Naturalist 55: 142-150.
Lesica, P. 1995. Demography of Astragalus scaphoides and effects of herbivory on population growth. Great Basin Naturalist 55: 142-150. Lesica, P., M.T. Lavin, and P.F. Stickney. 2022. Manual of Montana Vascular Plants, Second Edition. Fort Worth, TX: BRIT Press. viii + 779 p.
Lesica, P., M.T. Lavin, and P.F. Stickney. 2022. Manual of Montana Vascular Plants, Second Edition. Fort Worth, TX: BRIT Press. viii + 779 p. Vanderhorst, J.P. 1995. Sensitive plant survey in the Horse Prairie Creek drainage, Beaverhead County, Montana. Unpublished report to the Bureau of Land Management, Butte District. Montana Natural Heritage Program, Helena, Montana. 42 pp. plus appendices.
Vanderhorst, J.P. 1995. Sensitive plant survey in the Horse Prairie Creek drainage, Beaverhead County, Montana. Unpublished report to the Bureau of Land Management, Butte District. Montana Natural Heritage Program, Helena, Montana. 42 pp. plus appendices. Vanderhorst, J.P. and P. Lesica. 1995a. Sensitive plant survey of the Tendoy Mountains in the Beaverhead National Forest, Beaverhead County, Montana. Unpublished report to the Bureau of Land Management, Butte District. Montana Natural Heritage Program, Helena, MT. 59 pp. plus appendices.
Vanderhorst, J.P. and P. Lesica. 1995a. Sensitive plant survey of the Tendoy Mountains in the Beaverhead National Forest, Beaverhead County, Montana. Unpublished report to the Bureau of Land Management, Butte District. Montana Natural Heritage Program, Helena, MT. 59 pp. plus appendices.
- Web Search Engines for Articles on "Bitterroot Milkvetch"





