View in other NatureServe Network Field Guides
NatureServe
Montana
Utah
Wyoming
Idaho
Wisconsin
British Columbia
South Carolina
Yukon
California
New York
- Home - Other Field Guides
- Kingdom - - Animalia
- Phylum - Spiders, Insects, and Crustaceans - Arthropoda
- Class - Insects - Insecta
- Order - Sawflies / Wasps / Bees / Ants - Hymenoptera
- Family - Bumble, Honey, Carpenter, Stingless, & Orchid Bees - Apidae
- Species - Red-belted Bumble Bee - Bombus rufocinctus
Red-belted Bumble Bee - Bombus rufocinctus
Other Names:
Cullumanobombus rufocinctus
Native Species
Global Rank:
G5
State Rank:
S5
(see State Rank Reason below)
Agency Status
USFWS:
USFS:
BLM:
State Rank Reason (see State Rank above)
Species is widely distributed across Montana and relatively common within suitable habitat. Trend appears stable and does not appear to face significant threats.
General Description
For definitions and diagrams of bumble bee morphology please see the
Montana State Entomology Collection's Bumble Bee Morphology page. A short-tongued, small-sized bumble bee: queens 17-18 mm in length, workers 9-13 mm. Hair short and even; head very short, cheek distinctly shorter than wide; mid-leg basitarsus with the back far corner rounded, hind-leg tibia outer surface flat and hairless (except fringe) forming a pollen basket; many color patterns are expressed, but hair of T2 at front and near midline usually yellow, T1 yellow, T3 orange or black or both. Males 11-15 mm in length; eyes slightly enlarged and weakly convergent in the upper part, larger than eyes of any female bumble bee; antennae medium length, flagellum nearly 3X the length of scape; hair color pattern similar to queens and workers, but black band between wings on upper side of thorax intermixed with many yellow hairs (Colla et al. 2011, Koch et al. 2012, Williams et al 2014).
Phenology
Across the range, queens reported April to October, workers May to September, males June to September (Williams et al. 2014). In southern Ontario, queens reported May to September, workers June to September, males July to September (Colla and Dumesh 2010); in Utah, queens May to July, workers May to September, males July to October (Koch et al. 2012). In California, queens report late February to early October, workers late May to early October, males late June to late September (Thorp et al. 1983).
Diagnostic Characteristics
Please see the
Montana State Entomology Collection's Key to Female Bumble Bees in Montana. Females told from other Montana
Bombus by a combination of the outer surface of hind-leg concave and lacking hair (except fringe), pollen basket present; T2-3 with red or orange hair, sometimes with yellow intermixed at least in the middle; scutum predominantly yellow or pale yellow; face predominantly with black hair; cheek distinctly shorter than wide.
Species Range
Montana Range
Range Descriptions
 Native
Native
Recorded Montana Distribution
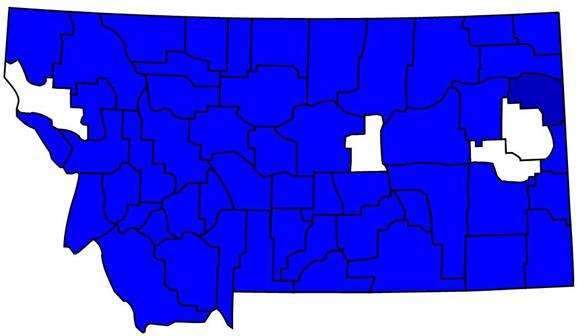
Click the map for additional distribution information.
Range Comments
Throughout the western United States west of the Great Plains from Canada south to southern California, Arizona, and New Mexico, and across southern Canada and adjacent northern tier of the United States from British Columbia to Quebec and New England (Williams et al. 2014). In Colorado, reported at elevations of 1600-4000 m, but most abundant below 2900 m (Macior 1974). Widespread and common throughout the range, persisting in many former collection sites in the east and possibly increasing in abundance at some localities (Colla and Packer 2008, Grixti et al. 2009, Colla et al. 2012, Williams et al. 2014).
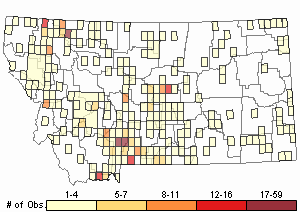
Observations in Montana Natural Heritage Program Database
Number of Observations: 710
(Click on the following maps and charts to see full sized version)
Map Help and Descriptions
Relative Density
Recency
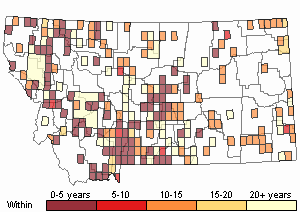
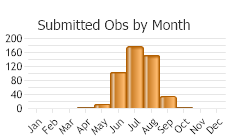
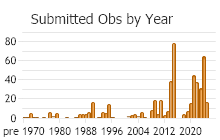
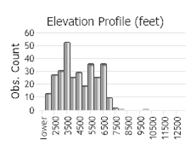 (Observations spanning multiple months or years are excluded from time charts)
(Observations spanning multiple months or years are excluded from time charts)
Habitat
Often near and in woodlands, urban parks and gardens in the east, also sagebrush steppe, aspen parkland, prairie grassland to alpine tundra, and commercial Vaccinium crops in the west (Macior 1974, Richards 1978, Ratti et al. 2008, Colla and Dumesh 2010, Cook et al. 2011). In sagebrush steppe, this species appears to become more abundant as sagebrush canopy cover declines (Cook et al. 2011).
Food Habits
Feeds on a wide diversity of flowers, including Apocynum, Arctium, Asclepias, Astragalus, Balsamorhiza, Brassica, Calypso, Campanula, Ceanothus, Centaurea, Chrysothamnus, Cichorium, Cirsium, Cleome, Coronilla, Daucus, Dodecatheon, Echium, Epilobium, Erigeron, Erysimum, Eupatorium, Fragaria, Frasera, Geranium, Grindelia, Haplopappus, Helianthus, Heracleum, Hypericum, Iris, Lactuca, Lupinus, Malus, Medicago, Melilotus, Mentha, Mertensia, Monarda, Onosmodium, Oxytropis, Pedicularis, Penstemon, Phacelia, Potentilla, Prunella, Rorippa, Rosa, Rubus, Sisyrinchium, Solidago, Symphyotrichum, Tanacetum, Taraxacum, Thermopsis, Trifolium, Triteleia, Vaccinium, Verbena, Vicia and Viguiera (Macior 1968, Beattie et al. 1973, Macior 1974, Ackerman 1981, Thorp et al. 1983, Mayer et al. 2000, Ratti et al. 2008, Wilson et al. 2010, Colla and Dumesh 2010, Colla et al. 2011, Koch et al. 2012, Miller-Struttmann and Galen 2014, Williams et al. 2014). An important pollinator in southern British Columbia of commercial Vaccinium crops such as highbush blueberry, and cranberry to a lesser extent (Ratti et al. 2008).
Reproductive Characteristics
Seems to favor nesting underground under some circumstances, but also commonly nests on the ground surface and above ground; (Plath 1934) reported a Vermont nest 2.6 m above ground in the clapboards of a house. When offered a choice between artificial underground and ground nests in the wooded foothills of the Rocky Mountains of southern Alberta, (Hobbs 1965b) documented 54.5% of 99 nests were constructed underground. In aspen parkland of southern Alberta, (Richards 1978) reported that 81.4% of 43 nests in one year and 58.2% of 67 nests the following year were built on the surface or above ground when queens were offered an equal choice of artificial nest sites. Nests initiated late May to mid July, with the majority started in June. Sometimes, conspecific queens attempt to take over established nests (Hobbs 1965b, Richards 1978). In first broods, the average number of eggs = 11.2 (range: 8-15), larvae = 10.2 (range: 7-13), and pupae (cocoons) = 10.4 (range: 7-15). Queens produce workers in 19-24 days after initiating nests. For second and third broods, 3-5 eggs (mean = 4.2) are laid per cell, in fourth and fifth broods 4-13 eggs (mean = 8.2) are laid per cell. Average numbers of cocoons produced in some colonies producing queens were 66-157; the largest colony produced 430 cocoons, at least 33 males, and 103 queens. During winter, queens construct hibernacula and hibernate in the ground at depths of 2.5-6.4 cm (Hobbs 1965b). Males maintain territories often centered on blooming shrubs, frequently changing perches and pursuing passing insects and birds in search of queens (O'Neill et al. 1991). Some nests are parasitized by the cuckoo bumble bees Bombus suckleyi, B. insularis, and B. flavidus (=fernaldae) (Hobbs 1965b).
Stewardship Responsibility
References
- Literature Cited AboveLegend:
 View Online Publication
View Online Publication Ackerman, J.D. 1981. Pollination biology of Calypso bulbosa var. occidentalis (Orchidaceae): a food-deception system. Madroño 28(3): 101-110.
Ackerman, J.D. 1981. Pollination biology of Calypso bulbosa var. occidentalis (Orchidaceae): a food-deception system. Madroño 28(3): 101-110. Beattie, A.J., D.E. Breedlove, and P.R. Ehrlich. 1973. The ecology of the pollinators and predators of Frasera speciosa. Ecology 54: 81-91.
Beattie, A.J., D.E. Breedlove, and P.R. Ehrlich. 1973. The ecology of the pollinators and predators of Frasera speciosa. Ecology 54: 81-91. Colla, S., L. Richardson, and P. Williams. 2011. Bumble bees of the eastern United States. Washington, DC: USDA Forest Service, Pollinator Partnership. 103 p.
Colla, S., L. Richardson, and P. Williams. 2011. Bumble bees of the eastern United States. Washington, DC: USDA Forest Service, Pollinator Partnership. 103 p. Colla, S.R. and L. Packer. 2008. Evidence for decline in eastern North American bumble bees (Hymenoptera: Apidae), with special focus on Bombus affinis Cresson. Biodiversity Conservation 17: 1379-1391.
Colla, S.R. and L. Packer. 2008. Evidence for decline in eastern North American bumble bees (Hymenoptera: Apidae), with special focus on Bombus affinis Cresson. Biodiversity Conservation 17: 1379-1391. Colla, S.R. and S. Dumesh. 2010. The bumble bees of southern Ontario: notes on natural history and distribution. Journal of the Entomological Society of Ontario 141:39-68.
Colla, S.R. and S. Dumesh. 2010. The bumble bees of southern Ontario: notes on natural history and distribution. Journal of the Entomological Society of Ontario 141:39-68. Colla, S.R., F. Gadallah, L. Richarson, D. Wagner, and L. Gall. 2012. Assessing declines of North American bumble bees (Bombus spp.) using museum specimens. Biodiversity and Conservation 21: 3585-3595.
Colla, S.R., F. Gadallah, L. Richarson, D. Wagner, and L. Gall. 2012. Assessing declines of North American bumble bees (Bombus spp.) using museum specimens. Biodiversity and Conservation 21: 3585-3595. Cook, S.P., S.A. Birch, F.W. Merickel, C.C. Lowe, and D. Page-Dumroese. 2011. Bumble bee (Hymenoptera: Apidae) community structure on two sagebrush steppe sites in southern Idaho. Pan-Pacific Entomologist 87(3): 161-171.
Cook, S.P., S.A. Birch, F.W. Merickel, C.C. Lowe, and D. Page-Dumroese. 2011. Bumble bee (Hymenoptera: Apidae) community structure on two sagebrush steppe sites in southern Idaho. Pan-Pacific Entomologist 87(3): 161-171. Grixti, J.C., L.T. Wong, S.A. Cameron, and C. Favret. 2009. Decline of bumble bees (Bombus) in the North American Midwest. Biological Conservation 142: 75-84.
Grixti, J.C., L.T. Wong, S.A. Cameron, and C. Favret. 2009. Decline of bumble bees (Bombus) in the North American Midwest. Biological Conservation 142: 75-84. Hobbs, G.A. 1965b. Ecology of species of Bombus Latr. (Hymenoptera: Apidae) in southern Alberta. III. Subgenus Cullumanobombus Vogt. Canadian Entomologist 97(12): 1293-1302.
Hobbs, G.A. 1965b. Ecology of species of Bombus Latr. (Hymenoptera: Apidae) in southern Alberta. III. Subgenus Cullumanobombus Vogt. Canadian Entomologist 97(12): 1293-1302. Koch, J., J. Strange, and P. Williams. 2012. Bumble bees of the western United States. Washington, DC: USDA Forest Service, Pollinator Partnership. 143 p.
Koch, J., J. Strange, and P. Williams. 2012. Bumble bees of the western United States. Washington, DC: USDA Forest Service, Pollinator Partnership. 143 p. Macior, L.M. 1968. Bombus (Hymenoptera, Apidae) queen foraging in relation to vernal pollination in Wisconsin. Ecology 49:20-25.
Macior, L.M. 1968. Bombus (Hymenoptera, Apidae) queen foraging in relation to vernal pollination in Wisconsin. Ecology 49:20-25. Macior, L.M. 1974. Pollination ecology of the Front Range of the Colorado Rocky Mountains. Melanderia 15: 1-59.
Macior, L.M. 1974. Pollination ecology of the Front Range of the Colorado Rocky Mountains. Melanderia 15: 1-59. Mayer, D.F., E.R. Miliczky, B.F. Finnigan, and C.A. Johnson. 2000. The bee fauna (Hymenoptera: Apoidea) of southeastern Washington. Journal of the Entomological Society of British Columbia 97: 25-31.
Mayer, D.F., E.R. Miliczky, B.F. Finnigan, and C.A. Johnson. 2000. The bee fauna (Hymenoptera: Apoidea) of southeastern Washington. Journal of the Entomological Society of British Columbia 97: 25-31. Miller-Struttmann, N.E. and C. Galen. 2014. High-altitude multi-taskers: bumble bee food plant use broadens along an altitudinal productivity gradient. Oecologia 176:1033-1045.
Miller-Struttmann, N.E. and C. Galen. 2014. High-altitude multi-taskers: bumble bee food plant use broadens along an altitudinal productivity gradient. Oecologia 176:1033-1045. O'Neill, K.M., H.E. Evans, and L.B. Bjostad. 1991. Territorial behaviour in males of three North American species of bumblebees (Hymenoptera: Apidae, Bombus). Canadian Journal of Zoology. 69(3) 604-613.
O'Neill, K.M., H.E. Evans, and L.B. Bjostad. 1991. Territorial behaviour in males of three North American species of bumblebees (Hymenoptera: Apidae, Bombus). Canadian Journal of Zoology. 69(3) 604-613. Plath, O.E. 1934. Bumblebees and their ways. New York, NY: Macmillan Company. 201 p.
Plath, O.E. 1934. Bumblebees and their ways. New York, NY: Macmillan Company. 201 p. Ratti, C.M., H.A. Higo, T.L. Griswold, and M.L. Winston. 2008. Bumble bees influence berry size in comercial Vaccinium spp. cultivation in British Columbia. Canadian Entomologist 140(3): 348-363.
Ratti, C.M., H.A. Higo, T.L. Griswold, and M.L. Winston. 2008. Bumble bees influence berry size in comercial Vaccinium spp. cultivation in British Columbia. Canadian Entomologist 140(3): 348-363. Richards, K.W. 1978. Nest site selection by bumble bees (Hymenoptera: Apidae) in southern Alberta. Canadian Entomologist 110(3): 301-318.
Richards, K.W. 1978. Nest site selection by bumble bees (Hymenoptera: Apidae) in southern Alberta. Canadian Entomologist 110(3): 301-318. Thorp, R.W., D.S. Horning, and L.L. Dunning. 1983. Bumble bees and cuckoo bumble bees of California (Hymenoptera: Apidae). Bulletin of the California Insect Survey 23:1-79.
Thorp, R.W., D.S. Horning, and L.L. Dunning. 1983. Bumble bees and cuckoo bumble bees of California (Hymenoptera: Apidae). Bulletin of the California Insect Survey 23:1-79. Williams, P., R. Thorp, L. Richardson, and S. Colla. 2014. Bumble Bees of North America. Princeton, NJ: Princeton University Press. 208 p.
Williams, P., R. Thorp, L. Richardson, and S. Colla. 2014. Bumble Bees of North America. Princeton, NJ: Princeton University Press. 208 p. Wilson, J.S., L.E. Wilson, L.D. Loftis, and T. Griswold. 2010. The montane bee fauna of north central Washington, USA, with floral associations. Western North American Naturalist 70(2): 198-207.
Wilson, J.S., L.E. Wilson, L.D. Loftis, and T. Griswold. 2010. The montane bee fauna of north central Washington, USA, with floral associations. Western North American Naturalist 70(2): 198-207.
- Additional ReferencesLegend:
 View Online Publication
View Online Publication
Do you know of a citation we're missing? Adhikari, S. 2018. Impacts of dryland farming systems on biodiversity, plant-insect interactions, and ecosystem services. Ph.D. Dissertation. Bozeman, MT: Montana State University. 207 p.
Adhikari, S. 2018. Impacts of dryland farming systems on biodiversity, plant-insect interactions, and ecosystem services. Ph.D. Dissertation. Bozeman, MT: Montana State University. 207 p. Adhikari, S., L.A. Burkle, K.M. O'Neill, C.M. Delphia, D.K. Weaver, and F.D. Menalled. 2019. Dryland organic farming partially offsets negative effects of highly simplified agricultural landscapes on forbs, bees, and bee-flower networks. Environmental Entomology 48(4): 826-835.
Adhikari, S., L.A. Burkle, K.M. O'Neill, C.M. Delphia, D.K. Weaver, and F.D. Menalled. 2019. Dryland organic farming partially offsets negative effects of highly simplified agricultural landscapes on forbs, bees, and bee-flower networks. Environmental Entomology 48(4): 826-835. Burkle L.A., M.P. Simanonok, J.S. Durney, J.A. Myers, and R.T. Belote. 2019. Wildfires influence abundance, diversity, and intraspecific and interspecific trait variation of native bees and flowering plants across burned and unburned landscapes. Frontiers in Ecology and Evolution 7(252):1-14.
Burkle L.A., M.P. Simanonok, J.S. Durney, J.A. Myers, and R.T. Belote. 2019. Wildfires influence abundance, diversity, and intraspecific and interspecific trait variation of native bees and flowering plants across burned and unburned landscapes. Frontiers in Ecology and Evolution 7(252):1-14. Delphia, C.M., Griswold, T., Reese, E.G., O'Neill, K.M., and Burkle, L.A. 2019. Checklist of bees (Hymenoptera: Apoidea) from small, diversified vegetable farms in south-western Montana. Biodiversity Data Journal: e30062
Delphia, C.M., Griswold, T., Reese, E.G., O'Neill, K.M., and Burkle, L.A. 2019. Checklist of bees (Hymenoptera: Apoidea) from small, diversified vegetable farms in south-western Montana. Biodiversity Data Journal: e30062 Dolan, A.C. 2016. Insects associated with Montana's huckleberry (Ericaceae: Vaccinium globulare) plants and the bumble bees (Hymenoptera: Apidae) of Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 160 p.
Dolan, A.C. 2016. Insects associated with Montana's huckleberry (Ericaceae: Vaccinium globulare) plants and the bumble bees (Hymenoptera: Apidae) of Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 160 p. Dolan, A.C., C.M. Delphia, K.M. O'Neill, and M.A. Ivie. 2017. Bumble Bees (Hymenoptera: Apidae) of Montana. Annals of the Entomological Society of America. 110(2): 129-144.
Dolan, A.C., C.M. Delphia, K.M. O'Neill, and M.A. Ivie. 2017. Bumble Bees (Hymenoptera: Apidae) of Montana. Annals of the Entomological Society of America. 110(2): 129-144. Fultz, J.E. 2005. Effects of shelterwood management on flower-visiting insects and their floral resources. M.Sc. Thesis. Bozeman, MT: Montana State University. 163 p.
Fultz, J.E. 2005. Effects of shelterwood management on flower-visiting insects and their floral resources. M.Sc. Thesis. Bozeman, MT: Montana State University. 163 p. Kearns, C.A. and J.D. Thomson. 2001. The Natural History of Bumble Bees. Boulder, CO. University Press of Colorado.
Kearns, C.A. and J.D. Thomson. 2001. The Natural History of Bumble Bees. Boulder, CO. University Press of Colorado. Reese, E.G., L.A. Burkle, C.M. Delphia, and T. Griswold. 2018. A list of bees from three locations in the Northern Rockies Ecoregion (NRE) of western Montana. Biodiversity Data Journal 6: e27161.
Reese, E.G., L.A. Burkle, C.M. Delphia, and T. Griswold. 2018. A list of bees from three locations in the Northern Rockies Ecoregion (NRE) of western Montana. Biodiversity Data Journal 6: e27161. Sater, S. 2022. The insects of Sevenmile Creek, a pictorial guide to their diversity and ecology. Undergraduate Thesis. Helena, MT: Carroll College. 242 p.
Sater, S. 2022. The insects of Sevenmile Creek, a pictorial guide to their diversity and ecology. Undergraduate Thesis. Helena, MT: Carroll College. 242 p. Simanonok, M. 2018. Plant-pollinator network assembly after wildfire. Ph.D. Dissertation. Bozeman, MT: Montana State University. 123 p.
Simanonok, M. 2018. Plant-pollinator network assembly after wildfire. Ph.D. Dissertation. Bozeman, MT: Montana State University. 123 p. Simanonok, M.P. and L.A. Burkle. 2019. Nesting success of wood-cavity-nesting bees declines with increasing time since wildfire. Ecology and Evolution 9:12436-12445.
Simanonok, M.P. and L.A. Burkle. 2019. Nesting success of wood-cavity-nesting bees declines with increasing time since wildfire. Ecology and Evolution 9:12436-12445.
- Web Search Engines for Articles on "Red-belted Bumble Bee"
- Additional Sources of Information Related to "Insects"





