View in other NatureServe Network Field Guides
NatureServe
Montana
Utah
Wyoming
Idaho
Wisconsin
British Columbia
South Carolina
Yukon
California
New York
Yellow Starthistle - Centaurea solstitialis
Other Names:
Yellow Star-thistle
State Rank Reason (see State Rank above)
Centaurea solstitialis is native to the Mediterranean region of Europe (Sheley and Petroff 1999). A conservation status rank is not applicable (SNA) because the plant is an exotic (non-native) in Montana that is not a suitable target for conservation activities.
General Description
PLANTS: An annual with erect, branched, and winged stems that grow 10–80 cm (Lesica et al. 2012). Plants are scabrous and gray-tomentose (Lesica et al. 2012).
LEAVES: Basal leaves form a rosette are are deeply lobed and approximately 5 cm wide and 20 cm long (Sheley and Petroff 1999). Stem leaves are entire, linear-oblanceolate, 5–15 cm long, short-petiolate, and have thin woolly hairs (Sheley and Petroff 2009; Lesica et al. 2012).
INFLORESCENCE: Corymbiform. Yellow flower heads are solitary on open leafy stems (FNA 2006). Involucres are ovoid and 13–17 mm tall (Lesica et al. 2012). The outer bracts are pale green and armed with a stout, straw-colored spine of 1-2 inches (2.5-5 cm) long (Sheley and Petroff 2009; Lesica et al. 2012). The spines radiate out into a star-shape. The inner bracts are lanceolate with swollen tips (Lesica et al. 2012). Corollas are 13–20 mm long. Pappus is absent or of white, unequal bristles of 2-4 mm long (FNA 2006). Fruits (cypselae) are 2–3 mm long and of two types: outer fruits are dark brown without a pappus and inner fruits are mottled white/light brown and with a pappus (FNA 2006).
Phenology
Flowering mostly from summer through autumn, but year-round in frost-free coastal areas (FNA 2006).
Diagnostic Characteristics
Yellow Starthistle is a winter annual with bright yellow flowers. The flower head (involucral) bracts are tipped with stout, straw-colored spines of 2.5-5.0 cm (1-2 inches) long (Sheley and Petroff 1999). The spines radiate from the flower head in a star shape. In the western U.S. and depending upon the growing conditions plants can be 10 cm to 1.5 meters tall (Sheley and Petroff 1999).
Species Range
Montana Range
Range Descriptions
 Non-native
Non-native
Range Comments
Yellow starthistle is native to the Mediterranean region of Europe (Sheley and Petroff 1999). It has been introduced into and occurs throughout most of the U.S. and southern Canada (Lesica et al. 2012).
It is believed to have arrived in North America as a contaminant with alfalfa seeds being shipped to California (Sheley and Petroff 1999). The earliest herbarium specimens are from California collected in the middle 1800’s. Seeds of yellow starthistle have been found in adobe brick made during the post-Mission Period (after 1824) in California (Sheley and Petroff 1999). Around 1900 it was reported near Bingen, Washington and began spreading into the grasslands of the Pacific Northwest in the 1920s (Sheley and Petroff 1999).
For maps and other distributional information on non-native species see:
Nonindigenous Aquatic Species Database from the U.S. Geological Survey
Invasive Species Habitat Tool (INHABIT) from the U.S. Geological Survey
Invasive Species Compendium from the Centre for Agriculture and Bioscience International (CABI)
EDDMapS Species Information EDDMapS Species Information
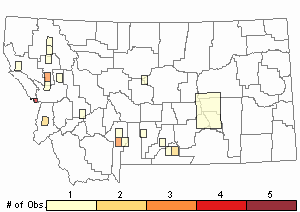
Observations in Montana Natural Heritage Program Database
Number of Observations: 29
(Click on the following maps and charts to see full sized version)
Map Help and Descriptions
Relative Density
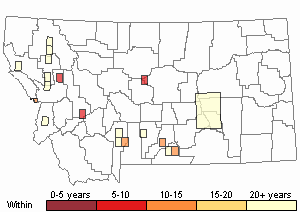
Recency
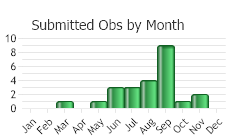
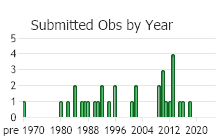
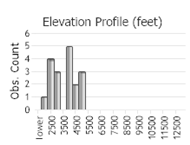 (Observations spanning multiple months or years are excluded from time charts)
(Observations spanning multiple months or years are excluded from time charts)
Habitat
Grasslands, rangeland, pastures, woodlands, roadsides, and wastelands (FNA 2006; Lesica et al. 2012).
Ecology
Yellow Starthistle’s competitive success is directly related to its rapid growth and ability to capture resources (Sheley and Petroff 1999). It is capable of invading rangelands throughout the western U.S (Sheley and Petroff 1999). In the Pacific Northwest rangelands with deep, loamy soils, south-facing slopes, and 30.4 to 63.5 cm (12 to 25 inches) of precipitation that peaks in spring or winter are most vulnerable. It competes well in areas dominated by perennial grasses, such as bluebunch wheatgrass (
Elymus spicatus), Idaho fescue (
Festuca idahoensis), and Sandberg bluegrass (
Poa secunda). It appears to not compete well with sagebrush (Artemisia spp.) unless the soil is already disturbed. However, Yellow Starthistle growth rates can decline as its density increases. Perennial grasses can compete against yellow starthistle invasions, particularly species that can initiate growth in the fall.
In deep-soiled rangelands Yellow Starthistle roots grow fast and deep (Sheley and Petroff 1999). Where cheatgrass (
Bromus tectorum) or other annual grasses dominate their rooting structure is more shallow. Yellow starthistle can outcompete annual grasses until its density limits growth or they can co-dominate. Where soils are shallow, cheatgrass has a competitive edge over Yellow Starthistle.
Whirlwinds, birds, humans, and other animals aid in the dispersal of Yellow Starthistle seeds. Seeds get dispersed by whirlwinds and human activities, such as driving or sticking to clothing (Sheley and Petroff 1999). Ring-necked Pheasants and California Quail consume whole seeds, which occasionally pass through their digestive tract in a viable form (Sheley and Petroff 1999). House and American Finches shell their seeds, which leaves them unviable (Sheley and Petroff 1999).
POLLINATORS The following animal species have been reported as pollinators of this plant species or its genus where their geographic ranges overlap:
Bombus vagans,
Bombus bifarius,
Bombus centralis,
Bombus fervidus,
Bombus rufocinctus,
Bombus ternarius,
Bombus terricola,
Bombus occidentalis,
Bombus griseocollis,
Bombus impatiens,
Bombus insularis, and
Bombus suckleyi (Thorp et al. 1983, Johnson 1986, Colla and Dumesh 2010, Koch et al. 2012, Williams et al. 2014, Tripoldi and Szalanski 2015).
Reproductive Characteristics
Yellow Starthistle produces two types of seeds: those with a parachute-like plume (i.e., a pappus of bristles) and those without. Most seeds have plumes and are dispersed by the wind. Plumeless seeds stay within the flower head and mostly disperse within two feet of the parent plant between November and February. Using an analogy, this builds up a slow moving invasion front.
LIFE CYCLE [Adapted from Sheley et al. in Sheley and Petroff 1999]
Yellow Starthistle are winter annuals, whereby seeds germinate in the fall. However, spring germination has been observed. Based on laboratory tests germination occurs at a fast rate. Plumed seeds germinate within 16 hours under optimal temperature (68 degrees Fahrenheit (20 degrees Celsius)) with unlimited moisture. After 48 hours, almost 75% of the plumed seeds and 44% of the plumeless seeds have germinated. This quick response implies that seeds can quickly take advantage of optimal conditions while many species cannot. Under dry or salty conditions germination rates are lower. Other studies found that about 95% of seeds are viable with 10% remaining viable for 10 years. In heavily infested stands the seedbank approaches 13% of total seed production with most being plumeless. A single plant has the potential to produce 150,000 seeds; however, a study near Walla Walla, Washington found plants produce 20-120 seeds. Seed production is dependent upon plant density and moisture.
Growth usually starts in the fall following precipitation. Frost heaving can reduce seedling survival. Seedlings transition to rosettes from March to early May. The rosette stage appears to be a vulnerable period. Studies have shown that 60-75% of rosettes may die by July due to moisture stress or self-thinning. From the rosette, a flowering stalk emerges. Mature plants can die from environmental stressors, but generally populations are stable from year to year. In early autumn, plants drop their leaves and die, leaving behind a silvery-grey skeleton with cottony-white seedheads. With autumn rains, seeds germinate, and the cycle repeats.
Management
See
Noxious Weed Education VideoSuccessful control requires integrating strategies to prevent new, eradicate or containing existing, and controlling large populations.
PREVENTION [Adapted from Sheley et al.
in Sheley and Petroff 1999]
Targeting actions that prevent or reduce seed production will hinder its ability to grow and spread. Spread can be reduced through many actions, such as, a) preventing vehicles from driving through and animals from grazing within infested areas, b) thoroughly washing the undercarriage of vehicles that have travel through infested areas, c) developing educational campaigns to teach people to not pick and transport the yellow flowers, and d) encouraging landowners to frequently monitor their land for new infestations and, when found to implement effective control methods.
BIOLOGICAL CONTROL [Adapted from Sheley et al.
in Sheley and Petroff 1999]
As of 1996 three weevils (
Bangasternus orientalis, Eustenopus villosus, Larinus curtus) and three flies (
Chaetorellia autralis, Urophora sirunaseva, Urophora jaculata) that attack Yellow Starthistle flower heads have been approved for released. They were released on plants in Canada and the Pacific Northwest, but apparently are not in Montana.
CHEMICAL CONTROL [Adapted from Sheley et al.
in Sheley and Petroff 1999]
Herbicides are effective, especially when properly managed with other tactics. The herbicide type and concentration, timing of chemical control, soil properties, and other factors will determine its effectiveness and impact to non-target species. Strict adherence to application requirements defined on the herbicide label will reduce risks to human and environmental health. Many herbicides must be applied by applicators with an Aquatic Pest Control license. Consult your County Extension Agent and/or Weed District for more information on herbicidal control.
Picloram at the 0.25 pound per acre rate can provide selective control when applied to seedlings, rosettes, or plants beginning to bolt without harm to well-established grasses. It cannot be used in sandy soils, near surface waters, or in areas with a high-water table.
2,4-D at the 0.5 pound per acre rate can be effectively used in riparian areas or around homesteads.
Clopyralid at the 1.2 pound per acre rate can be applied before plants produce flower buds and in areas with high water tables.
Glyphosate at the one pint per acre rate will kill all plants and can be used in revegetation projects that need to prepare an initial seedbed for seeding desirable vegetation.
PHYSICAL & CULTURAL CONTROLS [Adapted from Sheley et al.
in Sheley and Petroff 1999]
Hand-pulling is very effective because it is an annual. Pulled plants should be bagged and dessicated or preferably burned before disposing to ensure that seeds are not viable. Hand-pulling small infestations, around gardens and homesites, and within crop fields, riparian areas, and along waterways will eradicate or slowing its spread. Systematic surveys and repeated removal should be conducted every 2-4 weeks throughout the growing season.
Tilling and discing severs roots below the soil surface and will kill plants, but it should be done periodically through the growing season. This will deplete the seed source and lessen the severity for re-invasion. Once the ability for re-invasion has been controlled then desirable plants can be established.
Mowing is not effective because plants respond by growing more prostrate. Mowing in dry years might reduce seed production. Mowing before it rains will stimulate growth and increase seed production.
Burning populations will increase infestations. Fire releases nutrients and Yellow Starthistle seeds are quick to capture them, germinate, and grow.
Revegetation should follow any control method. A mix of perennial grass cover is best to interrupt the cycle of re-invasion because they compete strongly for light, nutrients, and water resources. This may require the use of particular cultivars of exotic and/or native grasses. Successful seeding will depend upon the particular grass species/cultivars, the density that establishes, environmental conditions, and the land manager’s ability to maintain grass vigor. It is recommended to not fertilize new grass seedings that are infested with yellow starthistle because it will increase the forb’s production.
GRAZING CONTROLS [Adapted from Sheley et al.
in Sheley and Petroff 1999]
Grazing and grazing management can be effective at controlling Yellow Starthistle. Cattle or sheep can suppress Yellow Starthistle if grazed when plants bolt, but before they develop spines. Prior to developing spines, the crude protein content of plants ranges from 5-10%. Repeated grazing is necessary to suppress growth, but the intensity and frequency should depend upon environmental conditions. In revegetated areas, the desirable rangeland plants should achieve full recovery before being grazed, to ensure they are most competitive against Yellow Starthistle.
Useful Links:Central and Eastern Montana Invasive Species TeamMontana Invasive Species websiteMontana Biological Weed Control Coordination ProjectMontana Department of Agriculture - Noxious WeedsMontana Weed Control AssociationMontana Weed Control Association Contacts WebpageMontana Fish, Wildlife, and Parks - Noxious WeedsMontana State University Integrated Pest Management ExtensionWeed Publications at Montana State University Extension - MontGuidesStewardship Responsibility
References
- Literature Cited AboveLegend:
 View Online Publication
View Online Publication Colla, S.R. and S. Dumesh. 2010. The bumble bees of southern Ontario: notes on natural history and distribution. Journal of the Entomological Society of Ontario 141:39-68.
Colla, S.R. and S. Dumesh. 2010. The bumble bees of southern Ontario: notes on natural history and distribution. Journal of the Entomological Society of Ontario 141:39-68. Flora of North America Editorial Committee. 2006. Flora of North America North of Mexico. Vol. 19. Magnoliophyta: Asteridae, part 6: Asteraceae, part 1. Oxford Univ. Press, New York. xxiv + 579 pp.
Flora of North America Editorial Committee. 2006. Flora of North America North of Mexico. Vol. 19. Magnoliophyta: Asteridae, part 6: Asteraceae, part 1. Oxford Univ. Press, New York. xxiv + 579 pp. Johnson, R.A. 1986. Intraspecific resource partitioning in the bumble bees Bombus ternarius and B. pennsylvanicus. Ecology 67:133-138.
Johnson, R.A. 1986. Intraspecific resource partitioning in the bumble bees Bombus ternarius and B. pennsylvanicus. Ecology 67:133-138. Koch, J., J. Strange, and P. Williams. 2012. Bumble bees of the western United States. Washington, DC: USDA Forest Service, Pollinator Partnership. 143 p.
Koch, J., J. Strange, and P. Williams. 2012. Bumble bees of the western United States. Washington, DC: USDA Forest Service, Pollinator Partnership. 143 p. Lesica, P., M.T. Lavin, and P.F. Stickney. 2012. Manual of Montana Vascular Plants. Fort Worth, TX: BRIT Press. viii + 771 p.
Lesica, P., M.T. Lavin, and P.F. Stickney. 2012. Manual of Montana Vascular Plants. Fort Worth, TX: BRIT Press. viii + 771 p. Sheley, Roger, and Janet Petroff. 1999. Biology and Management of Noxious Rangeland Weeds. Oregon State University Press, Corvallis, Oregon.
Sheley, Roger, and Janet Petroff. 1999. Biology and Management of Noxious Rangeland Weeds. Oregon State University Press, Corvallis, Oregon. Thorp, R.W., D.S. Horning, and L.L. Dunning. 1983. Bumble bees and cuckoo bumble bees of California (Hymenoptera: Apidae). Bulletin of the California Insect Survey 23:1-79.
Thorp, R.W., D.S. Horning, and L.L. Dunning. 1983. Bumble bees and cuckoo bumble bees of California (Hymenoptera: Apidae). Bulletin of the California Insect Survey 23:1-79. Tripoldi, A.D. and A.L. Szalanski. 2015. The bumble bees (Hymenoptera: Apidae: Bombus) of Arkansas, fifty years later. Journal of Melittology 50: doi: http://dx.doi.org/10.17161/jom.v0i50.4834
Tripoldi, A.D. and A.L. Szalanski. 2015. The bumble bees (Hymenoptera: Apidae: Bombus) of Arkansas, fifty years later. Journal of Melittology 50: doi: http://dx.doi.org/10.17161/jom.v0i50.4834 Williams, P., R. Thorp, L. Richardson, and S. Colla. 2014. Bumble Bees of North America. Princeton, NJ: Princeton University Press. 208 p.
Williams, P., R. Thorp, L. Richardson, and S. Colla. 2014. Bumble Bees of North America. Princeton, NJ: Princeton University Press. 208 p.
- Additional ReferencesLegend:
 View Online Publication
View Online Publication
Do you know of a citation we're missing? Dillard, S.L. 2019. Restoring semi-arid lands with microtopography. M.Sc. Thesis. Bpzeman, MT: Montana State University. 97 p.
Dillard, S.L. 2019. Restoring semi-arid lands with microtopography. M.Sc. Thesis. Bpzeman, MT: Montana State University. 97 p. Lehnhoff, E.A., L.J.Rew, J.M. Mangold, T. Seipel, and D. Ragen.2019. Integrated management of Cheatgrass (Bromus tectorum) with sheep grazing and herbicide. Agronomy 9, 315
Lehnhoff, E.A., L.J.Rew, J.M. Mangold, T. Seipel, and D. Ragen.2019. Integrated management of Cheatgrass (Bromus tectorum) with sheep grazing and herbicide. Agronomy 9, 315 Lesica, P., M.T. Lavin, and P.F. Stickney. 2022. Manual of Montana Vascular Plants, Second Edition. Fort Worth, TX: BRIT Press. viii + 779 p.
Lesica, P., M.T. Lavin, and P.F. Stickney. 2022. Manual of Montana Vascular Plants, Second Edition. Fort Worth, TX: BRIT Press. viii + 779 p. Olliff, Tom, Roy Renkin, Craig McClure, Paul Miller, Dave Price, Dan Reinhart, and Jennifer Whipple. 2001. Managing A Complex Exotic Vegetation Program in Yellowstone National Park.
Olliff, Tom, Roy Renkin, Craig McClure, Paul Miller, Dave Price, Dan Reinhart, and Jennifer Whipple. 2001. Managing A Complex Exotic Vegetation Program in Yellowstone National Park.
- Web Search Engines for Articles on "Yellow Starthistle"





