View in other NatureServe Network Field Guides
NatureServe
Montana
Utah
Wyoming
Idaho
Wisconsin
British Columbia
South Carolina
Yukon
California
New York
Salt Cedar - Tamarix ramosissima
Other Names:
Tamarisk, Salt-cedar, Saltcedar,
[including hybrids with] Tamarix chinensis
State Rank Reason (see State Rank above)
Tamarix ramosissima is a plant native to the Middle East and introduced into North America (Jacobs and Sing 2012). A conservation status rank is not applicable (SNA) because the plant is an exotic (non-native) in Montana that is not a suitable target for conservation activities.
NOTE: There is a discrepancy between the number of observations and counties represented in the herbaria (Consortium of Pacific Northwest Herbaria; www.pnwherbaria.org) versus the Montana Natural Heritage Program databases, which indicates a problem. It is important to document new occurrences with nice quality plant specimens of which some should be deposited in our State herbaria (University of Montana, Montana State University, Montana State University-Billings). Depositing specimens in the herbaria allows identifications to be confirmed, provides a central location for educating and sharing information, and allows specimens to be studied for genetics, morphology, and ecology.
General Description
PLANTS: Deciduous large shrubs from 1-5 meters tall. Numerous slender stems arise from a branched caudex. Bark is thin, reddish-brown, but with age becomes furrowed and ridged. Source: Lesica et al. 2012; Jacobs and Sing 2012.
LEAVES: Leaves are arranged alternately. Leaves resemble cedar because they are small, 1-2mm, scale-like, and clasping the branches; however, unlike cedar leaves are deciduous. Leaves are succulent and broadly lanceolate. On the underside of leaves are salt-secreting glands. Source: Lesica et al. 2012; Jacobs and Sing 2012.
INFLORESCENCE: Small pink (occasionally white) flowers are arranged in narrow, drooping clusters (spike-like, bracteate racemes) of 15–40 mm long. Flowers are perfect, regular, hypogenous. The 5 sepals are separate, and 1 mm or less long. The 5 pink petals are separate, ovate, about 2 mm long. Flower have 5 stamens and 3-4 stigmas. Source: Lesica et al. 2012; Jacobs and Sing 2012.
Tamarisk is from the Arabic tamr which refers to a tree with dark bark (Gaskin in FNA 2015). Tamarix is derived from the Tambre River which in ancient times was referred to as Tamaris River in Spain (Jacobs and Sing 2012).
Phenology
Flowers from spring through September, sometimes October (Jacobs and Sing 2012).
Diagnostic Characteristics
5-Stamen Tamarisk (
Tamarix chinensis) and Salt Cedar (
Tamarix ramosissima) are genetically distinct species that do not naturally overlap in native geographical ranges. 5-Stamen Tamarisk occurs from western China to Japan which is east of central China. Salt Cedar occurs from Turkey to Japan, which is west of central China. In North America where both species have been introduced, hybrids have been found (Gaskin
in FNA 2015). Hybrids occur in Montana as well. Montana's plants best fit
Tamarix ramosissima (Lesica et al. 2012). Hybridized specimens as well as sterile collections of Salt Cedar have led to many misidentifications (Gaskin
in FNA 2015). Directions for how to dissect flowers and find the nectar disc and other characteristics is described by Gaskin
in FNA 2015.
Salt Cedar -
Tamarix ramosissima, exotic and noxious:
* Wispy shrubby growth form that often grows in riparian areas.
* Deciduous leaves that are very small, grey-green, and are appressed to twigs.
* Short racemes of tiny pink flowers that can cover the shrub for most of the growing season.
* Margins of sepals are denticulate.
* Stamen filaments originate from the edge of the nectar disc.
* Montana herbarium specimens at time of publication appear to better represent Salt Cedar (Lesica et al. 2012).
* It will rarely hybridize with Athel Tamarisk> (
Tamarix aphylla) (Gaskin
in FNA 2015).
5-Stamen Tamarisk -
Tamarix chinensis, exotic and noxious:
* Wispy shrubby growth form that often grows in riparian areas.
* Deciduous leaves that are very small, grey-green, and appressed to twigs.
* Short racemes of tiny pink flowers that can cover the shrub for most of the growing season.
* Margins of sepals entire.
* Some or all filaments originate from below the nectar disc.
* Reported to occur in Montana by Gaskin
in FNA 2015; however, University of Montana and Montana State herbarium specimens appear to better resemble Salt Cedar (Lesica et al. 2012).
* It will rarely hybridize with Athel Tamarisk (
Tamarix aphylla).
Rocky Mountain Juniper -
Juniperus scopulorum, native:
* An evergreen shrub or small tree that can grow in some riparian areas and in upland habitats.
* Leaves are small, dark green, and appressed to twigs. First year needles are short and needle-like.
* Plants are a true conifer with separate male and female plants. Male plants produce ephemeral cones and female plants produce glaucous-blue berries.
Western Redcedar –
Thuja plicata, native and desirable:
* An evergreen large tree that grow in some riparian areas and other moist habitats.
* Leaves are small, dark green, glossy, and appressed to twigs. Twigs are flattened and drooping.
* Plants are a true conifer that bear female and male cones.
Cedar, exotic and cultivated:
* An evergreen shrub and true conifer (cultivated) planted as row hedges and in yards.
Species Range
Montana Range
Range Descriptions
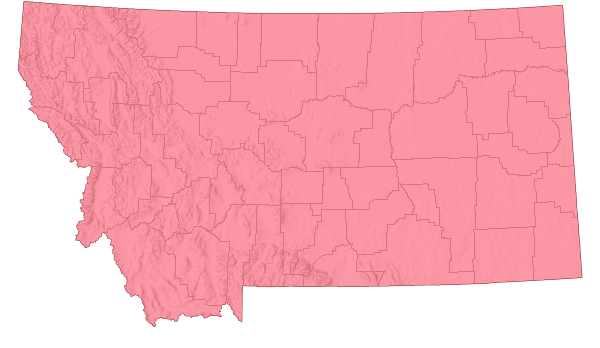
 Non-native
Non-native
Range Comments
Salt Cedar was introduced to North America from the Middle East in the early 1800s. It occurs in most of temperate, western North America (Lesica et al. 2012). This shrub was introduced for use as a garden ornamental, windbreak, and erosion control.
Herbarium specimens document its presence in Big Horn County, Montana in 1971 (Specimens posted as of February 21, 2019 at the Consortium of Pacific Northwest Herbarium portal, www.pnwherbaria.org). However, other reports indicate it may have been in Montana from the 1960s in Treasure and Rosebud Counties (Swenson et al. 1982).
For maps and other distributional information on non-native species see:
Nonindigenous Aquatic Species Database from the U.S. Geological Survey
Invasive Species Habitat Tool (INHABIT) from the U.S. Geological Survey
Invasive Species Compendium from the Centre for Agriculture and Bioscience International (CABI)
EDDMapS Species Information EDDMapS Species Information
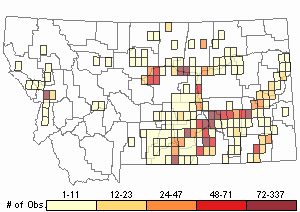
Observations in Montana Natural Heritage Program Database
Number of Observations: 7506
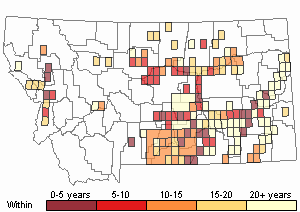
(Click on the following maps and charts to see full sized version)
Map Help and Descriptions
Relative Density
Recency
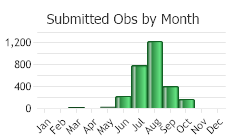
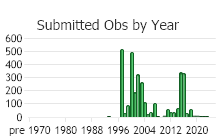
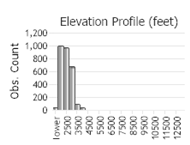 (Observations spanning multiple months or years are excluded from time charts)
(Observations spanning multiple months or years are excluded from time charts)
Habitat
Salt Cedar grows along streams, stream terraces, saline meadows along waterways, lakes, irrigation ditches, and reservoirs in the plains zone of Montana (Lesica et. al 2012). It grows well in areas that exhibit repeated cycles of inundation and drawdown, particularly where there is disturbance, depressional wetlands, or stock ponds, and where prolonged inundation is absent (Lesica and Miles 2004).
Ecology
Salt Cedar is an early successional species adapted to flood and drought conditions. Its root system grows deep to draw from ground water and from unsaturated soil (Lesica and Miles 2004). However, cottonwoods and native willows cannot survive when groundwater levels drop to below their root depths.
Salt Cedar often colonizes the same habitats that can support cottonwood (
Populus spp.) forests or willow (
Salix spp.) thickets (Lesica and Miles 2004). It colonizes moist, disturbed habitats near water bodies, such as point bars and drawdown zones (Lesica and Miles 2004). However, willows and cottonwoods are more adapted to streams that rise in response to snow melt, which generally time well with the production of their short-lived seeds (Lesica and Miles 2004). Salt Cedar produces seed throughout the growing season which allows its to colonize streams that develop little snow accumulation, such as the Missouri River tributaries east of the Mussellshell River (Lesica and Miles 2004). It can establish and live longer by colonizing areas of declining hydrology that do get some subirrigation. Studies from Fort Peck Reservoir suggested that the Western Wheatgrass habitat type is susceptible to invasion by Salt Cedar (Lesica and Miles 2004). Western Wheatgrass habitats that have disturbance, depressional wetlands, or stock ponds are most susceptible to invasion. The consequence would result in the gradually decline of perennial grass cover as Salt Cedar shrubs increase (Lesica and Miles 2004). In elevations not influenced by the Fort Peck Reservoir, Salt Cedar was uncommon (Lesica and Miles 2004).
Salt Cedar grows best in full sun. In Montana plants growing in the shade of cottonwoods were a little shorter, grew fewer live stems, and had a lower growth rates when compared with Salt Cedar growing in full sun (Lesica and Miles 2001). Further the study concluded that Salt Cedar growing in the shade did not initiate stem growth as much as shrubs growing in the sun (Lesica and Miles 2001). In Montana Salt Cedar seedlings have a slower growth rate when compared with cottonwood seedlings, and shrubs dieback at the end of the growing season (Lesica and Miles 2001). By the time both plants are 10 years old, undamaged cottonwood trees will overtop and shade Salt Cedar shrubs. Therefore, cottonwood species have a competitive advantage over Salt Cedar in Montana (Lesica and Miles 2001). In eastern Montana Salt Cedar appears to be opportunistic, colonizing early successional habitat that has also been manipulated by humans, but is not capable of acheiving the height and leaf-area of populations in the southwestern U.S. (Lesica and Mills 2001). It appears that Montana populations will remain as a understory shrub, declining as native woody plants mature (Lesica and Mills 2001). Management can curtail Salt Cedar by promoting actions that favor native cottonwoods and willows, such as reducing livestock damage and maintaining flood flows that are timed with cottonwood seed dispersal (Lesica and Mills 2001).
Some early studies (pre-1990) indicate that Salt Cedar uses more water than native riparian species, and it was believed to increase water losses through evapotranspiration. However, recent studies using improved methods for estimating evapotranspiration have shown that Salt Cedar does not use more water than native vegetation (Jacobs and Sing 2012).
SALINITYSalt Cedar is more deeply rooted and tolerant of salinity than most native plants. Salt Cedar can pull water from saline soils by accumulating more salt in their leaves. Plants excrete the excess salt through glands on the underside of the leaves. Leaves can become encrusted with salt crystals. As leaf little builds up so does the salt encrusted in them. About two inches of salt-encrusted leaf litter can prevent the establishment of other plants.
FLOODING [Adapted from Lesica and Miles 2004; Jacobs and Sing 2012]
Seedlings can withstand inundation for up to 4 weeks. Mature plants can withstand flooding for 70 days while root crowns can withstand flooding for 98 days. Generally, plants cannot survive inundation for 3 months or longer. Older plants generally occur along the high water mark. Saplings usually occur in flood zones. At the Fort Peck Reservoir, Salt Cedar plants germinate well in the drawdown zones, but die by the time they become 5 years. This is because these zones will eventually become inundated, and plants cannot tolerate 3 months of inundation. Scouring can prevent the build up of Salt Cedar populations. In comparison to cottonwood species, Salt Cedar has a longer window where seeds are viable, allowing it to take advantage of flooding and flow released by dams.
WILDLIFE INTERACTIONS [Adapted from Jacobs and Sing 2012]
Salt Cedar has limited usefulness for animals. Dense stands are unsuitable for many mammals, reptiles, amphibians, and certain groups of birds, such as timber drillers and cavity nesters. However, the southwestern willow flycatcher, an endangered bird, nests in Salt Cedar in some regions of the western U.S. Mixed stands of native vegetation and Salt Cedar can provide good habitat for many species.
Beaver select native species over Salt Cedar by a ratio of 10:1. This allows Salt Cedar to grow while removing the native woody vegetation.
POLLINATORS The following animal species have been reported as pollinators of this plant species or its genus where their geographic ranges overlap:
Bombus vagans (Colla and Dumesh 2010).
Reproductive Characteristics
Plants reproduce by seed and from a caudex that grows secondary roots that branch and sprout vegetative shoots (clones).
FRUIT [Adapted from Jacobs and Sing 2012]
Fruit is a capsule. Capsule is 3–4 mm long with many seeds. Seeds are black with an apical tuft of hairs. The tuft helps the seed float in air or on water.
ROOTS [Adapted from Jacobs and Sing 2012]
Plants form an extensive root system. The taproot grows deep to intersect groundwater. Adventitious buds occur on the vascular cambium of stems and branches and develop adventitious roots. Therefore, stems can grow from buds on the root crown or from buds on the roots. It has been found that the lateral roots can grow up to 6 meters long and the taproot can grow 3.5 feet deep.
LIFE CYCLE [Adapted from Jacobs and Sing 2012]
Established plants in Montana often re-sprout from root crowns in the spring. Stems can grow quickly, up to 4 meters in a season. In early spring buds break to develop into branches or leaves. Plants flower beginning the first year after establishment; although, viable seed won’t develop until the plant is 3 years old. Maturity is reached at 3 years of age. Flowers bloom from May through September, or October if conditions are appropriate. Seeds primarily disperse by wind. Seeds can also be dispersed by boats, in water ballast, on boat trailers, and on fisherman’s waders.
A mature Salt Cedar plant can produce half a million seeds each year. Seeds are produced throughout the growing season when soil moisture is enough for maintaining active plant metabolism. Seeds are short-lived, usually germinating within 24 hours of dispersal under suitable conditions. Under ideal summer conditions seeds may remain viable for up to 45 days before desiccating. Under ideal winter conditions seed viability is longer, up to 130 days. Upon contact with moisture seeds germinate quickly, often forming carpets of seedlings. Seedlings totaling 800 per square meter have been counted. Typically, less than 10% will survive their first year.
Leaves drop in October (though in warm climates plant may be more evergreen). Come winter stems often dieback to the ground. The life expectancy of individual shrubs is 20 years.
Management
An integrated vegetative management approach provides the best long-term control, and requires that land-use objectives and a desired plant community be identified (Shelly et al.
in Sheley and Petroff 1999). Once identified an integrated weed management strategy that promotes a weed-resistant plant community and serves other land-use objectives such as livestock forage, wildlife habitat, or recreation can be developed.
* Where willows and cottonwood grow, beavers might need to be controlled in order to allow native shrubs to mature and provide the shade that will reduce Salt Cedar from establishing (Lesica and Miles 2004).
* Areas that exhibit repeated cycles of inundation and drawdown will be susceptible to Salt Cedar invasion, particularly where there is disturbance, depressional wetlands, or stock ponds and where prolonged inundation is absent (Lesica and Miles 2004).
* Management can curtail Salt Cedar by promoting actions that favor native cottonwoods and willows, such as reducing livestock damage and maintaining flood flows that are timed with cottonwood seed dispersal (Lesica and Mills 2001).
PREVENTION [Adapted from Jacobs and Sing 2012]
A
2025 Montana Woody Invasive Species Statewide Management Plan identifies management priorities, summarizes treatment and removal best practices, and outlines short- and long-term implementation objectives for this species as well as Russian Olive (
Elaeagnus angustifolia) and Common Buckthorn (
Rhamnus cathartica) (Ricklefs et al. 2025).
In riparian areas that are not infested with Salt Cedar, surveys should be conducted every 3 years in order to detect new populations. In riparian areas within 25 miles of known populations, intensive surveys should be conducted to detect new populations and prevent establishment. Learn how to accurately identify Salt Cedar in order to detect occurrences and know where to implement control(s) to eradicate new populations.
PHYSICAL and CULTURAL CONTROLS [Adapted from Jacobs and Sing 2012]
Hand-pulling is not usually effective because a huge number of seedlings germinate when conditions are good. Most seedlings won’t survive. Those that become established are too difficult to hand-pull.
Mowing,
Chaining,
Cutting,
Prescribed Burningand
Tilling are not effective because these stimulate re-sprouting and require follow-up treatments.
Controlled flooding for greater than 3 months at reservoirs and stock ponds may reduce Salt Cedar populations, as well as native trees and shrubs. After flooding re-vegetation may be necessary.
Natural flooding cycles on free-flowing streams is believed to control Salt Cedar establishment. It can scour seedlings, deposits of salt, and accumulations of leaf litter. Manipulating water releases from dams that mimics natural flows may favor establishment of native woody riparian vegetation and select again Salt Cedar. Salt Cedar seedlings of up to 5 weeks old are more susceptible to summer flooding then are established plants. Prolonged flooding of 3 or more months can kill Salt Cedar (Lesica and Miles 2004).
Re-vegetation that uses native plants adapted to the particular site conditions creates shade that will negatively impact Salt Cedar. Guidance on seeding and planting techniques can be found in the Riparian Forest Buffer (Code 391) and Riparian Herbaceous Cover (Code 390) practice standards and specifications.
BIOLOGICAL CONTROL [Adapted from Jacobs and Sing 2012]
Biological control has been restricted due to the perceived threat posed to the federally threatened Southwestern sub-species of the Willow Flycatcher (
Empidonax traillii).
The
Northern Tamarisk Beetle (Diorhabda carinulata) which originates from northwestern China and eastern Kazakhstan has been released in Montana. The adult and larval stages feed mostly on young leaves and less on mature leaves. They can occasionally feed on the epidermis of twigs and first-year shoots which can cause the tip and of the twig to dry-up and fall off. In Montana the beetles have not been successful possibly due to predation or environment factors of spring drought or spring flooding.
CHEMICAL CONTROL [Adapted from Jacobs and Sing 2012]
Herbicides can be effective, especially when properly integrated with intensive pasture management. The herbicide type and concentration, application time and method, environmental constraints, land use practices, local regulations, and other factors will determine its effectiveness and impact to non-target species. Strict adherence to application requirements defined on the herbicide label will reduce risks to human and environmental health. Consult your County Extension Agent and/or Weed District for information on herbicidal control. Chemical information is also available at Greenbook.
Basal Bark Treatment: For scattered shrubs, a single low volume basal bark treatment using a 30% solution of trycolpyr mixed with a commercially available basal bark oi, number 1 or 2 dielsel fuel, or kerosene is high effective and practical. The circumference of all stems from the basal root crown upwards to 18 inches should be sprayed until thoroughly wet but not dripping. The treatment can be used except when the ground is frozen or flooded. It will take 1-2 years for the herbicide to move through the plant, particularly its roots, and kill it. This chemical can be used along non-irrigation ditch banks, seasonally dry wetlands, and transitional areas between wetlands and uplands where surface water is not present.
Cut Stump Treatment: In areas where stems can be cut, triclopyr or glyphosate can be applied directly to the cut stump. It must be applied within 1 hour to prevent the plant from sprouting. The herbicide can be applied to wet the wood and surrounding cambium. The treatment can be used except when the ground is frozen or flooded. Stems that are removed should be burned in order to make sure they don’t re-sprout.
Foliar Treatment: Where stands are dense, imazapyr (2 quarts per acre) or imazapyr plus glyphosate (1 quart per acre each) are the most effective for spraying Salt Cedar foliage. Applications in late summer or early fall (August to September) are most effective. Spot treatments can be followed by a 1 percent solution of imazapyr with a surfactant.
GRAZING CONTROL [Adapted from Jacobs and Sing 2012]
Salt Cedar is nutritionally poor for cattle, sheep, and goats; however, they will eat it. Livestock tend to select native species over Salt Cedar, which then favors the establishment of Salt Cedar. When Salt Cedar is eaten, cattle graze on young sprouts in the early spring. Confining domesticated animals to graze Salt Cedar can work to remove above ground foliage, but will encourage sprouting. Boer Goats have been used to intensively graze Salt Cedar. Intensive grazing following by a chemical application will improve the effectiveness of the herbicide.
In areas were cottonwood and willows grow, livestock management should encourage the regeneration and growth of these native woody species because their shade will later discourage Salt Cedar (Lesica and Miles 2004).
COSTS:
Modeling habitat suitability for the species under future mid-range climate scenarios indicates a dramatic 224% increase in the area of optimally suitable habitat (Nelson et al. 2026). Combined reported mitigation costs for Salt Cedar and Russian Olive through 2025 are approximately $5.5 million, total mitigation costs are currently estimated at $28 million for Salt Cedar alone, and total mitigation costs for Salt Cedar under future climate scenarios are expected to rise to $103 million (Nelson et al. 2026).
ASSISTANCE:Montana's Salt Cedar Task Force is led by Rachel Frost who can be contacted at: (406) 454-0056 or
mrcdc@macdnet.orgUseful Links:Central and Eastern Montana Invasive Species TeamMontana Invasive Species websiteMontana Biological Weed Control Coordination ProjectMontana Department of Agriculture - Noxious WeedsMontana Weed Control AssociationMontana Weed Control Association Contacts WebpageMontana Fish, Wildlife, and Parks - Noxious WeedsMontana State University Integrated Pest Management ExtensionWeed Publications at Montana State University Extension - MontGuidesStewardship Responsibility
Threats or Limiting Factors
Salt Cedar has successfully colonized, established, or dominated along rivers, streams, lakes, irrigation ditches, and reservoirs and altered their hydrology. High densities of Salt Cedar can congest river channels and create potential flood hazards. Salt Cedar reduces channel widths by decreasing the water velocity and thereby increasing sediment deposition.
Research from the southwestern U.S. indicates that native vegetation may decrease in Salt Cedar infested areas, but research in Montana showed no differences in plant community composition or species richness in infested versus non-infested areas. Instead, differences in species composition may be more associated with water flow regimes which are altered by dams and encourage Salt Cedar invasions. In comparison to the Southwest U.S. Salt Cedar infestations in Montana are younger and may not have had time to negatively affect plant communities.
Where dense Salt Cedar can prevent native (and other exotic) species from re-establishing because their fallen leaves have a high salt content.
Salt Cedar can reduce small mammal diversity, alter avian composition in riparian communities.
It increases evapo-transpiration-mediated water loss from riparian systems, although losses now appear to be similar to that of native communities.
References
- Literature Cited AboveLegend:
 View Online Publication
View Online Publication Colla, S.R. and S. Dumesh. 2010. The bumble bees of southern Ontario: notes on natural history and distribution. Journal of the Entomological Society of Ontario 141:39-68.
Colla, S.R. and S. Dumesh. 2010. The bumble bees of southern Ontario: notes on natural history and distribution. Journal of the Entomological Society of Ontario 141:39-68. Flora of North America Editorial Committee, eds. 2015. Flora of North America north of Mexico, Volume 6. Magnoliophyta: Cucurbitaceae to Droseraceae. Oxford University Press, Inc. New York.
Flora of North America Editorial Committee, eds. 2015. Flora of North America north of Mexico, Volume 6. Magnoliophyta: Cucurbitaceae to Droseraceae. Oxford University Press, Inc. New York. Jacobs, Jim and Sharlene Sing. 2012. Ecology and Management of Saltcedar (Tamarix spp). September. U.S. Department of Agriculture, Natural Resources Conservation Service, Bozeman, Montana.
Jacobs, Jim and Sharlene Sing. 2012. Ecology and Management of Saltcedar (Tamarix spp). September. U.S. Department of Agriculture, Natural Resources Conservation Service, Bozeman, Montana. Lesica, P. and S. Miles. 2001. Tamarisk growth at the northern margin of its naturalized range in Montana, USA. Wetlands, 21(2): 240-246.
Lesica, P. and S. Miles. 2001. Tamarisk growth at the northern margin of its naturalized range in Montana, USA. Wetlands, 21(2): 240-246. Lesica, P., M.T. Lavin, and P.F. Stickney. 2012. Manual of Montana Vascular Plants. Fort Worth, TX: BRIT Press. viii + 771 p.
Lesica, P., M.T. Lavin, and P.F. Stickney. 2012. Manual of Montana Vascular Plants. Fort Worth, TX: BRIT Press. viii + 771 p. Lesica, Peter and Scott Miles. 2004. Ecological strategies for managing tamarisk on the C.M. Russell National Wildlife Refuge, Montana, USA. Biological Conservation 119 (2004), pages 535-543.
Lesica, Peter and Scott Miles. 2004. Ecological strategies for managing tamarisk on the C.M. Russell National Wildlife Refuge, Montana, USA. Biological Conservation 119 (2004), pages 535-543. Nelson, N.M., D. Whited, and B. Burkholder. 2026. Current and future expected costs of woody invasive species in Montana - Russian Olive (Eleagnus angustifolia), Tamarix (Tamarix chinensis, T. ramosissima, and hybrids thereof) and Common Buckthorn (Rhamnus cathartica). Flathead Lake Biological Station, University of Montana, Polson, Montana. 36 p.
Nelson, N.M., D. Whited, and B. Burkholder. 2026. Current and future expected costs of woody invasive species in Montana - Russian Olive (Eleagnus angustifolia), Tamarix (Tamarix chinensis, T. ramosissima, and hybrids thereof) and Common Buckthorn (Rhamnus cathartica). Flathead Lake Biological Station, University of Montana, Polson, Montana. 36 p. Ricklefs, S., J. Riddle, and E. Bjorklund. 2025. Montana woody invasive species statewide management plan. Prepared for Montana Noxious Weed Trust Fund, Montana Department of Agriculture, Helena, Montana. 127 pp.
Ricklefs, S., J. Riddle, and E. Bjorklund. 2025. Montana woody invasive species statewide management plan. Prepared for Montana Noxious Weed Trust Fund, Montana Department of Agriculture, Helena, Montana. 127 pp. Sheley, Roger, and Janet Petroff. 1999. Biology and Management of Noxious Rangeland Weeds. Oregon State University Press, Corvallis, Oregon.
Sheley, Roger, and Janet Petroff. 1999. Biology and Management of Noxious Rangeland Weeds. Oregon State University Press, Corvallis, Oregon.
- Additional ReferencesLegend:
 View Online Publication
View Online Publication
Do you know of a citation we're missing? Boggs, K. W. 1984. Succession in riparian communities of the lower Yellowstone River, Montana. M.S. Thesis. Montana State University, Bozeman, 107 pp.
Boggs, K. W. 1984. Succession in riparian communities of the lower Yellowstone River, Montana. M.S. Thesis. Montana State University, Bozeman, 107 pp. Eggers, M.J.S. 2005. Riparian vegetation of the Montana Yellowstone and cattle grazing impacts thereon. M.Sc. Thesis. Montana State University, Bozeman, MT. 125 p.
Eggers, M.J.S. 2005. Riparian vegetation of the Montana Yellowstone and cattle grazing impacts thereon. M.Sc. Thesis. Montana State University, Bozeman, MT. 125 p. Fritzen, D.E. 1995. Ecology and behavior of Mule Deer on the Rosebud Coal Mine, Montana. Ph.D. Dissertation. Bozeman, MT: Montana State University. 143 p.
Fritzen, D.E. 1995. Ecology and behavior of Mule Deer on the Rosebud Coal Mine, Montana. Ph.D. Dissertation. Bozeman, MT: Montana State University. 143 p. Lesica, P., M.T. Lavin, and P.F. Stickney. 2022. Manual of Montana Vascular Plants, Second Edition. Fort Worth, TX: BRIT Press. viii + 779 p.
Lesica, P., M.T. Lavin, and P.F. Stickney. 2022. Manual of Montana Vascular Plants, Second Edition. Fort Worth, TX: BRIT Press. viii + 779 p. Swenson, J.E., P. Hendricks, and A. Farjon. 1982. Arrival and occurrence of Tamarix chinensis (Tamarisk) along the Yellowstone River in Treasure and Rosebud County, Montana. Proceedings of the Montana Academy of Science 41:67-70.
Swenson, J.E., P. Hendricks, and A. Farjon. 1982. Arrival and occurrence of Tamarix chinensis (Tamarisk) along the Yellowstone River in Treasure and Rosebud County, Montana. Proceedings of the Montana Academy of Science 41:67-70.
- Web Search Engines for Articles on "Salt Cedar"





