View in other NatureServe Network Field Guides
NatureServe
Montana
Utah
Wyoming
Idaho
Wisconsin
British Columbia
South Carolina
Yukon
California
New York
Field Bindweed - Convolvulus arvensis
State Rank Reason (see State Rank above)
Convolvulus arvensis is native to the Mediterranea region and was introduced into North America (Piper in Sheley and Petroff 1999). A conservation status rank is not applicable (SNA) because the plant is an exotic (non-native) in Montana that is not a suitable target for conservation activities.
NOTE: There is a discrepancy between the number of observations and counties represented in the herbaria (Consortium of Pacific Northwest Herbaria; www.pnwherbaria.org) versus the Montana Natural Heritage Program databases, which indicates a problem. It is important to document new occurrences with nice quality plant specimens of which some should be deposited in our State herbaria (University of Montana, Montana State University, Montana State University-Billings). Depositing specimens in the herbaria allows identifications to be confirmed, provides a central location for educating and sharing information, and allows specimens to be studied for genetics, morphology, and ecology.
General Description
PLANTS: A rhizomatous, perennial vine. Stems are pubescent, prostrate or twining, branched at the base, and grow 1-6 feet long. Stems are corrugated longitudinally with a thick cuticle. Sources: Jacob 2007; Lesica et al. 2012.
LEAVES: Alternately arranged with blades 1-5 cm long and petioles (leaf stem) 5-25 mm long. Blades are arrowhead-shaped with rounded tips and 2 basal lobes that point backwards and outwards (sagittate). Margins are smooth (entire). Sources: Jacob 2007; Lesica et al. 2012.
INFLORESCENCE: Flowers are solitary and whitish, turning pinkish with age. The 5 petals are fused to form a funnel-shaped flower with almost indistinguishable lobes. The funnel-shape widens upward and flares outward at the edges. The 5 sepals are green with pink margins and overlap. Flower stem (peduncle) is 1–5 cm long with a pair of small bracts just below the flower. Source: Lesica et al. 2012.
The scientific name was given by Linnaeus in 1753. Convolvulus comes from a Latin verb “to roll together” or “to entwine” and arvensis is Latin adjective for “of the field” (Jacobs 2007). Since the time of Ancient Greeks this plant has been given at least 84 names (Jacobs 2007). The Romans’ name meant “a large worm that wraps itself in vines”. “Byndweeded” was first applied in England during the 1500s.
Phenology
Plants emerge in spring, flower throughout the summer with appropriate moisture, and die-back with freezing temperatures. See LIFE CYCLE / REPRODUCTIVE CHARACTERISTICS.
Diagnostic Characteristics
Whether by scientific name, common name, or looks these plants are often confused with each other:
Field Bindweed -
Convolvulus arvensis, exotic and Noxious:
* Vine in the Morning Glory Family (Convolvulcaeae) with a perennial life cycle.
* Flowers are showy, whitish, and solitary.
* Corolla is smaller, 1-3 cm long AND the bracts below the calyx are linear.
* Narrowly arrow-like leaves, but tips are mostly rounded.
Hedge False Bindweed -
Calystegia sepium, exotic and undesirable:
* Vine in the Morning Glory Family (Convolvulcaeae) with a perennial life cycle.
* Flowers are showy, whitish, and solitary.
* Corolla is larger, 3-7 cm long AND the bracts below the calyx also enclose the calyx.
* Heart-shaped or arrow-like leaves, but tips are pointed.
Black Bindweed -
Polygonum convolvulus, exotic and undesirable:
* Vine in the Buckwheat Family (Polygonaceae) with an annual life cycle.
* Flowers are very small (not showy), green, and clustered in leaf axils.
* Leaves are more heart-shaped (broader), but with pointed tips AND basal lobes.
Several
Ipomoea species resemble Field Bindweed, but have an annual habit, capitate stigma, longer sepals, and a blue or purple corolla.
Species Range
Montana Range
Range Descriptions
 Non-native
Non-native
Range Comments
Field Bindweed is a native of Eurasia and was introduced to North America in the 1730s (Wiese and Phillips 1976). It spread westward, reaching Pennsylvania by 1812, Kansas by 1877, and all the western states by 1900 (Whitesides 1979). It was first reported in California near San Francisco in 1838 and now infests 1.8 million acres in the state (Rosenthal 1983). It is spread by sowing contaminated crop seed, planting nursery stock containing convolvulus roots, and seed or plant parts carried by animals and humans (Swan 1980).
Adhikari et al. (2020) modeled the predicted future distribution of this and other weed species across the major road network in Montana under predicted future climate scenarios and found that area of predicted future habitat suitability is likely to increase.
For maps and other distributional information on non-native species see:
Nonindigenous Aquatic Species Database from the U.S. Geological Survey
Invasive Species Habitat Tool (INHABIT) from the U.S. Geological Survey
Invasive Species Compendium from the Centre for Agriculture and Bioscience International (CABI)
EDDMapS Species Information EDDMapS Species Information
Observations in Montana Natural Heritage Program Database
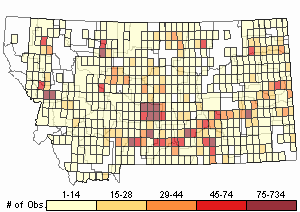
Number of Observations: 7541
(Click on the following maps and charts to see full sized version)
Map Help and Descriptions
Relative Density
Recency
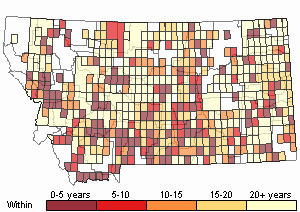
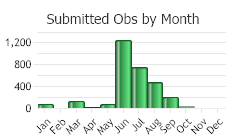
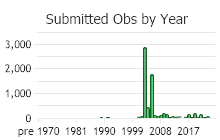
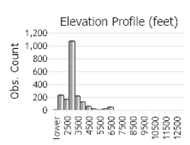 (Observations spanning multiple months or years are excluded from time charts)
(Observations spanning multiple months or years are excluded from time charts)
Habitat
Field Bindweed grows in cultivated fields, pastures, gardens, lawns, roadsides, railroad beds, and waste places (Jacobs 2007). It tolerates drought (Jacobs 2007). It grows best in rich, fertile soils that are dry to moderately moist, but will persist on poor, gravelly soils (Jacobs 2007). In Montana it is found in the plains and valleys (Lesica et al. 2012).
Ecology
POLLINATION and DISPERSAL Source: Jacob 2007.
Flowers persist for 1-day, produce nectar, and are pollinated by honey and bumble bees in the Family Hymenoptera, butterflies, and moths.
Potential and Known Pollinators:
The following animal species have been reported as pollinators of this plant species or its genus where their geographic ranges overlap:
Bombus bifarius and
Bombus griseocollis (Thorp et al. 1983).
Animals that ingest plants can transport and deposit seeds long-distances. In Migratory animals the hard, impermeable seed coat allows seeds to remain viable in the stomach for up to 144 hours.
Field Bindweed seeds can contaminate seed-crops and have intentionally been sold in flower packets as “wild morning glory” in the U.S. for use in ornamental gardens, as ground cover, or in hanging baskets. Seeds adhere to vehicles and field equipment, muddy boots, hoofs, and other objects, allowing plants to spread long-distances. Farm equipment can spread roots and rhizomes within and between fields.
RESTORATION Source: Jacob 2007.
Field Bindweed stems have been found to accumulate more than 3,800 milligrams (mg) of chromium, 1,500 mg of cadmium, and 560 mg of copper perkilogam of dry tissue. It may be a suitable plan for phytoremediation of soils that are comtamined with heavy metals.
CULTURAL Source: Jacob 2007.
Field Bindweed has been used as a purgative (root), laxative, and gynecological aid, to stop bleeding, to stimulate bile flow, and as a medicine for spider bites.
The Okanagan-Colville people in British Columbia and Washington made a rope out of the twining stems.
Reproductive Characteristics
Field Bindweed reproduces by seed and rhizome.
FLOWERS Sources: Jacob 2007; Lesica et al. 2012.
* The calyx is green with pink margins, 3–5 mm long, composed of 5 obovate sepals that overlap.
* The corolla is white or pinkish, becoming pink with age. The 5 fused petals are 15–25 mm long.
* The 5 stamens are unequal in length, attach to the corolla base between lobes, and are shorter than the petals (included).
* A single pistil has one style and a single 2-chambered ovary. The fruit is the capsule. The single capsule is 4–7 mm long, and produces 1-2 seeds in each chamber(locule).
* A seed is 3-sided with one flat and two rounded sides, 3-5 mm long, and weigh about 10 milligrams. The seed coat is hard, impermeable, dull brownish-gray, and coarsely-rough.
ROOTS AND RHIZOMES [Adapted from Jacobs et al. 2007]
The primary root is a whitish, skinny taproot that can grow 2-10 feet deep. Lateral roots grow from the taproot and from adventitious buds at the base of the stem. Buds on the lateral roots give rise to whitish rhizomes. Rhizomes are skinny and grow extensively within the upper 60 cm of the soil profile. The entire roots system of an individual plant can occupy an area of 6 meters in diameter and 9 meters in depth. As the root and rhizomes ages their color will darken (stained by the soil).
LIFE CYCLE [Adapted from Jacobs et al. 2007]
Seeds germinate throughout the growing season with adequate moisture, but peak from late spring to early summer. Stems grow when day temperatures reach 60 degrees Fahrenheit and night temperatures remain above 35 degree Fahrenheit.
In early spring buds on the lateral roots give rise to rhizomes. When rhizomes reach the soil surface a new crown develops. Each crown develops into an individual plant (a clone of its parent). When day temperatures reach 60 degrees Fahrenheit and night temperatures remain above 35 degree Fahrenheit, rhizomatous growth is rapid. From photosynthesis plants accumulate starch in their roots from August to September. The starch is converted to sugars in the roots where it acts like an “anti-freeze” protecting cells from freezing during winter. Carbyohydrate levels in the roots are lowest in May when the plant is actively growing and highest in October when it is actively storing. Once the plant is hardened in the fall, roots and rhizomes can survive soil temperatures as low as 21 degrees Fahrenheit. Roots and rhizomes occurring closer to the soil surface are injured when the ground freezes.
Plants flower in late June through fall, as long as conditions remain favorable. Each flower persists for 1 day. Flowers produce nectar and are pollinated by insects and are not capable of self-pollinating.
Seed set is most successful under dry, sunny weather and on dry-calcareous soils. Seed set may fail on heavy, poorly-drained soils during rainy periods. Seeds can germinate 10-15 days after pollination when their moisture content is 80%. Seeds can remain impermeable to water for about 23-25 days after pollination. Fresh seeds are from 87% to 99% viable and 5% to 25% can germinate, and 60% to 80% can become dormant due to the seed coat’s impermeability. Dormancy is broken by scarification or treating with sulfuric acid or ethyl alcohol. Over-wintering reduces permeability by 30%. In the soil seed can remain viable for at least 20 years. Seeds stored at room temperature can remain viable for 50 years.
Plants dieback to the root crown when temperatures approach freezing.
Seeds disperse near to the parent plant or are transported by water, animals, or man-made vectors. See ECOLOGY section.
Management
Persistent control over many years is required to significantly suppress Field Bindweed because of its extensive root system. Combining techniques of cultural, mechanical, biological, and chemical controls will best exhaust the nutrients in its root system (but must be designed to the site’s specific conditions). An integrated vegetative management approach provides the best long-term control, and requires that land-use objectives and a desired plant community be identified (Shelly et al.
in Sheley and Petroff 1999). Once identified an integrated weed management strategy can be developed that can promote a weed-resistant plant community and serves other land-use objectives such as livestock forage, wildlife habitat, or recreation.
PREVENTION [Adapted from Sheley and Petroff 1999]
Preventing the establishment of Field Bindweed can be accomplished by many practices:
* Learn how to accurately identify Field Bindweed in order to detect occurrences and know where to implement control methods.
* Prevent vehicles from driving through and animals from grazing within infested areas.
* Thoroughly wash the undercarriage of vehicles and wheels in a designated area before moving to uninfested areas.
* Frequently monitor for new plants, and when found implement effective control methods.
* Maintain proper grazing management that creates resilience to noxious weed invasion.
* Do not pick the flowers or transport plants. Where possible, contribute to or develop educational campaigns to help eradicate or reduce Field Bindweed populations.
PHYSICAL and CULTURAL CONTROLS [Adapted from Jacobs et al. 2007]
Hand-pulling is effective for young populations and small confined spaces if done several times each season to starve the root-system. Hand-pulling plants in combination with other control methods will likely be more successful at longer-term removal of the population.
Tilling fragments the rhizomes allowing Field Bindweed to increase in abundance. A chisel plow encourages re-growth of Field Bindweed. However, tilling every 8-12 days after re-growth for 3-5 consecutive years will reduce root reserves and deplete the seed bank in the soil. Tilling the plant when in flower can be more effective because root reserves of carbohydrates and nitrogen are at their lowest. Upon re-growth, a sweep plow can be used to remove top growth and keep it on the soil surface. After tilling, fields should be re-vegetated as soon as is practical. Appropriate herbicides could be used at the appropriate time to suppress or eradicate Field Bindweed.
Revegetating land with competitive, locally adapted, and desirable forbs and grasses or crops will suppress Field Bindweed populations. Field Bindweed grows in full sunlight and can be suppressed by plants that actively and densely grow in the spring, creating shade. Nonetheless, Field Bindweed will twine upwards around other plants, decreasing their competitive advantage. Establishing competitive, perennial grasses and forbs on disturbed land followed by a prescribed grazing management plant to maintain grass vigor will suppress Field Bindweed and reduce spread by seed. Planting with appropriate native plants is highly encouraged. Refer to Montana Plant Materials Technical Note 46, Seeding Rates for Conservation Species for Montana, and Extension Bulletin EB0019, Dryland Pasture Species for Montana and Wyoming for possible species selection and seeding rates.
Mowing is not generally effective because plants grow along the ground and remain under the height of the blade. Timing mowing with flowering (to decrease seed production) is difficult because plants flower throughout the season and flowers are viable for one-day. Mowing can spread the biological control mite
Aceria malherbae.
CHEMICAL CONTROLS [Adapted from Jacob 2007]
Biotypes of Field Bindweeds have different tolerances to herbicides. Herbicides can suppress Field Bindweed, will not be 100% effective, and will be more effective when properly integrated with an intensive pasture management. The herbicide type and concentration, application time and method, environmental constraints, land use practices, local regulations, and other factors will determine its effectiveness and impact to non-target species. Strict adherence to application requirements defined on the herbicide label will reduce risks to human and environmental health. Consult your County Extension Agent and/or Weed District for information on herbicidal control. Chemical information is also available at
Greenbook.
Plants growing in high light and low humidity environments have a thicker leaf cuticle and will absorb less herbicide than leaves of plants grown in a low light and high humidity environment. Herbicide absorption decreases when applied to dusty leaves in fields.
Picloram (1 quart per acre) or in combination with
Glyphosate,
2,4-D, or
Dicamba can control Field Bindweed for at least one year.
Picloram is a restricted-use herbicide because it is persistent, yet mobile in the soil, and can contaminate water and has a long-residual activity.
Many chemical types and techniques for controlling Field Bindweed in cropland are available by consulting your County’s Farm Service agency, Weed Coordinators, and MSU extension service (Jacobs 2007).
BIOLOGICAL CONTROLS [Adapted from Jacobs et al. 2007]
Bindweed Gall Mite (
Aceria malherbae) is native from central and southern Europe to northern Africa. Adults are minute (need a microscope), soft-bodied, and worm-like. They have ring-like body segments and 2-pairs of legs on their head and thorax. The nymps look similar to adults but lack external genitalia. Adults and nymphys are destructive to Field Bindweed. When attacked galls form on actively growing leaves, petioles, and stem tips. Infected leaves fold or twist upward along the mid-vein. Attacked buds don’t elongate, but will form a compact cluster of stunted leaves. Bindweed Gall Mites have multiple generations per year. Adults and nymphs over-winter on the root buds of Field Bindweed.
Bindweed Gall Mites can be collected as adults or nymphs during the growing season by selecting stems with galls and then wrapping them around actively growing stem tips of Field Bindweed at other sites. Transplanting releases in the spring or early summer will provides more time for establishment. Mowing can spread this mite. Herbicide treatment and tilling can hurt it.
Field Bindweed Moth (
Tyta luctuosa) is native from Europe to southern Scandinavia, Asia east to Turkistan and south into India, and Northern Africa. First generation adult moths emerge in May and are active until June. Second generation adult moths are active from July to September. Larvae are caterpillar-like and feed on the plant from May through September. At night they feed on Field Bindweed flowers and leaves and during the day they feed on plant litter. Thus, it is the larvae that damage Field Bindweed plants. Both adults and larvae over-winter on root buds of Field Bindweed. Numerous releases in 2002 in the U.S. have not successfully established this insect.
Bindweed Gall Mite and Field Bindweed Moth can also attack other bindweed plants in the genus Calystegia. Information on attacks to non-target species should be shared with the Montana Natural Program Heritage Program Botanist.GRAZING CONTROLS [Adapted from Jacobs et al. 2007]
Horses may develop an intestinal fibrosis because of the alkaloid pseudotropine that is found in Field Bindweed. Cattle, sheep, and goats will eat the leaves and stems of Field Bindweed. Chickens and hogs will eat leaves, stems, exposed roots and rhizomes, and crowns. However ingestion will cause Field Bindweed to spread. It has been found that the hard seed coat can keep the seeds variable for 144 hours in migratory animals.
Useful Links:Central and Eastern Montana Invasive Species TeamMontana Invasive Species websiteMontana Biological Weed Control Coordination ProjectMontana Department of Agriculture - Noxious WeedsMontana Weed Control AssociationMontana Weed Control Association Contacts WebpageMontana Fish, Wildlife, and Parks - Noxious WeedsMontana State University Integrated Pest Management ExtensionWeed Publications at Montana State University Extension - MontGuidesStewardship Responsibility
References
- Literature Cited AboveLegend:
 View Online Publication
View Online Publication Adhikari, A., L.J. Rew, K.P. Mainali, S. Adhikari, and B.D. Maxwell. 2020. Future distribution of invasive weed species across the major road network in the state of Montana, USA. Regional Environmental Change 20(60):1-14. https://doi.org/10.1007/s10113-020-01647-0
Adhikari, A., L.J. Rew, K.P. Mainali, S. Adhikari, and B.D. Maxwell. 2020. Future distribution of invasive weed species across the major road network in the state of Montana, USA. Regional Environmental Change 20(60):1-14. https://doi.org/10.1007/s10113-020-01647-0 Lesica, P., M.T. Lavin, and P.F. Stickney. 2012. Manual of Montana Vascular Plants. Fort Worth, TX: BRIT Press. viii + 771 p.
Lesica, P., M.T. Lavin, and P.F. Stickney. 2012. Manual of Montana Vascular Plants. Fort Worth, TX: BRIT Press. viii + 771 p. Sheley, Roger, and Janet Petroff. 1999. Biology and Management of Noxious Rangeland Weeds. Oregon State University Press, Corvallis, Oregon.
Sheley, Roger, and Janet Petroff. 1999. Biology and Management of Noxious Rangeland Weeds. Oregon State University Press, Corvallis, Oregon.
- Additional ReferencesLegend:
 View Online Publication
View Online Publication
Do you know of a citation we're missing? Adhikari, S. 2018. Impacts of dryland farming systems on biodiversity, plant-insect interactions, and ecosystem services. Ph.D. Dissertation. Bozeman, MT: Montana State University. 207 p.
Adhikari, S. 2018. Impacts of dryland farming systems on biodiversity, plant-insect interactions, and ecosystem services. Ph.D. Dissertation. Bozeman, MT: Montana State University. 207 p. Best, K. F. 1963. Note on the extent of lateral spread of field bindweed. Can. J. Plant Sci. 43: 230-232.
Best, K. F. 1963. Note on the extent of lateral spread of field bindweed. Can. J. Plant Sci. 43: 230-232. Cronquist, A., A.H. Holmgren, N.H. Holmgren, J.L. Reveal, and P.K. Holmgren. 1984. Intermountain Flora: Vascular Plants of the Intermountain West, U.S.A. Vol. 4, Subclass Asteridae (except Asteraceae). Bronx, NY: New York Botanical Garden. 573 pp.
Cronquist, A., A.H. Holmgren, N.H. Holmgren, J.L. Reveal, and P.K. Holmgren. 1984. Intermountain Flora: Vascular Plants of the Intermountain West, U.S.A. Vol. 4, Subclass Asteridae (except Asteraceae). Bronx, NY: New York Botanical Garden. 573 pp. DeGennaro, F. P., and S. C. Weller. 1982. Field bindweed biotype studies in Indiana. Proc. N. Cent. Weed Control Conf. 37: 47-48.
DeGennaro, F. P., and S. C. Weller. 1982. Field bindweed biotype studies in Indiana. Proc. N. Cent. Weed Control Conf. 37: 47-48. DeGennaro, F. P., and S. C. Weller. 1984b. Growth and reproductive characteristics of field bindweed (Convolvulus arvensis) biotypes. Weed Sci. 32: 525-528.
DeGennaro, F. P., and S. C. Weller. 1984b. Growth and reproductive characteristics of field bindweed (Convolvulus arvensis) biotypes. Weed Sci. 32: 525-528. DuBois, K.L. 1979. An inventory of the avifauna in the Long Pines of Southeastern Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 113 p.
DuBois, K.L. 1979. An inventory of the avifauna in the Long Pines of Southeastern Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 113 p. Eggers, M.J.S. 2005. Riparian vegetation of the Montana Yellowstone and cattle grazing impacts thereon. M.Sc. Thesis. Montana State University, Bozeman, MT. 125 p.
Eggers, M.J.S. 2005. Riparian vegetation of the Montana Yellowstone and cattle grazing impacts thereon. M.Sc. Thesis. Montana State University, Bozeman, MT. 125 p. Fritzen, D.E. 1995. Ecology and behavior of Mule Deer on the Rosebud Coal Mine, Montana. Ph.D. Dissertation. Bozeman, MT: Montana State University. 143 p.
Fritzen, D.E. 1995. Ecology and behavior of Mule Deer on the Rosebud Coal Mine, Montana. Ph.D. Dissertation. Bozeman, MT: Montana State University. 143 p. Hickman, M. V., and D. G. Swan. 1983. Comparison of rhizomes to lateral roots of field bindweed (Convolvulus arvensis L.) for seasonal variation in establishment. Proc. W. Soc. Weed Sci. 36: 77-81.
Hickman, M. V., and D. G. Swan. 1983. Comparison of rhizomes to lateral roots of field bindweed (Convolvulus arvensis L.) for seasonal variation in establishment. Proc. W. Soc. Weed Sci. 36: 77-81. Holm, L.G., P. Donald, J.V. Pancho, and J.P. Herberger. 1977. The World's Worst Weeds: Distribution and Biology. The University Press of Hawaii: Honolulu, Hawaii. 609 pp.
Holm, L.G., P. Donald, J.V. Pancho, and J.P. Herberger. 1977. The World's Worst Weeds: Distribution and Biology. The University Press of Hawaii: Honolulu, Hawaii. 609 pp. Jordon, L. S., and J. L. Jordan. 1982. Effect of pre-chilling on Convolvulus arvensis L. seed coat and germination. Ann. Bot. 49: 421-423.
Jordon, L. S., and J. L. Jordan. 1982. Effect of pre-chilling on Convolvulus arvensis L. seed coat and germination. Ann. Bot. 49: 421-423. King, L.A. 1980. Effects of topsoiling and other reclamation practices on nonseeded species establishment on surface mined land at Colstrip, Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 129 p.
King, L.A. 1980. Effects of topsoiling and other reclamation practices on nonseeded species establishment on surface mined land at Colstrip, Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 129 p. Lesica, P., M.T. Lavin, and P.F. Stickney. 2022. Manual of Montana Vascular Plants, Second Edition. Fort Worth, TX: BRIT Press. viii + 779 p.
Lesica, P., M.T. Lavin, and P.F. Stickney. 2022. Manual of Montana Vascular Plants, Second Edition. Fort Worth, TX: BRIT Press. viii + 779 p. Maxwell, B.D. 1984. Changes in an infested plant community after an application of picloram, the effect of glyphosate on bud dormancy, the effect of pulling and the fuel potential of leafy spurge (Euphorbia esula L.). M.Sc. Thesis. Bozeman, MT: Montana State University. 73 p.
Maxwell, B.D. 1984. Changes in an infested plant community after an application of picloram, the effect of glyphosate on bud dormancy, the effect of pulling and the fuel potential of leafy spurge (Euphorbia esula L.). M.Sc. Thesis. Bozeman, MT: Montana State University. 73 p. Meier, G.A. 1997. The colonization of Montana roadsides by native and exotic plants. M.Sc. Thesis. Bozeman, MT: Montana State University. 45 p.
Meier, G.A. 1997. The colonization of Montana roadsides by native and exotic plants. M.Sc. Thesis. Bozeman, MT: Montana State University. 45 p. Meyer, L. J. 1978. The influence of environment on growth and control of field bindweed. Proc. N. Cent. Weed Control Conf. 33: 141-142.
Meyer, L. J. 1978. The influence of environment on growth and control of field bindweed. Proc. N. Cent. Weed Control Conf. 33: 141-142. Munz, P.A., and D.D. Keck. 1973. A California flora and supplement. Univ. California Press, Berkeley, CA.
Munz, P.A., and D.D. Keck. 1973. A California flora and supplement. Univ. California Press, Berkeley, CA. Peterson, J.G. 1969. The food habits and summer distribution of juvenile sage grouse in central Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 39 p.
Peterson, J.G. 1969. The food habits and summer distribution of juvenile sage grouse in central Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 39 p. Phillips, W. M. 1978. Field bindweed: the weed and the problem. Proc. N. Cent. Weed Control Conf. 33: 140-141.
Phillips, W. M. 1978. Field bindweed: the weed and the problem. Proc. N. Cent. Weed Control Conf. 33: 140-141. Quire, R.L. 2013. The sagebrush steppe of Montana and southeastern Idaho shows evidence of high native plant diversity, stability, and resistance to the detrimental effects of nonnative plant species. M.Sc. Thesis. Bozeman, MT: Montana State University. 124 p.
Quire, R.L. 2013. The sagebrush steppe of Montana and southeastern Idaho shows evidence of high native plant diversity, stability, and resistance to the detrimental effects of nonnative plant species. M.Sc. Thesis. Bozeman, MT: Montana State University. 124 p. Rosenthal, S. S. 1983. Field bindweed in California: extent and cost of infestation. Calif. Agric. 37: 16-17.
Rosenthal, S. S. 1983. Field bindweed in California: extent and cost of infestation. Calif. Agric. 37: 16-17. Rundquist, V.M. 1973. Avian ecology on stock ponds in two vegetational types in north-central Montana. Ph.D. Dissertation. Bozeman, MT: Montana State University. 112 p.
Rundquist, V.M. 1973. Avian ecology on stock ponds in two vegetational types in north-central Montana. Ph.D. Dissertation. Bozeman, MT: Montana State University. 112 p. Sater, S. 2022. The insects of Sevenmile Creek, a pictorial guide to their diversity and ecology. Undergraduate Thesis. Helena, MT: Carroll College. 242 p.
Sater, S. 2022. The insects of Sevenmile Creek, a pictorial guide to their diversity and ecology. Undergraduate Thesis. Helena, MT: Carroll College. 242 p. Seipel, T.F. 2006. Plant species diversity in the sagebrush steppe of Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 87 p.
Seipel, T.F. 2006. Plant species diversity in the sagebrush steppe of Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 87 p. Skinner, K.F. 1995. Plant and grasshopper community composition: indicators & interactions across three spatial scales. M.Sc. Thesis. Bozeman, MT: Montana State University. 144 p.
Skinner, K.F. 1995. Plant and grasshopper community composition: indicators & interactions across three spatial scales. M.Sc. Thesis. Bozeman, MT: Montana State University. 144 p. Swan, D. G. 1980. Field bindweed, Convolvulus arvensis L. Washington State Univ. Coll. of Agric. Research Center Bull. 0888. Pullman, Washington.
Swan, D. G. 1980. Field bindweed, Convolvulus arvensis L. Washington State Univ. Coll. of Agric. Research Center Bull. 0888. Pullman, Washington. Timmons, J. 1949. Duration of viability of bindweed seed under field conditions and experimental results in the control of bindweed seedlings. Agron. J. 41: 130-133.
Timmons, J. 1949. Duration of viability of bindweed seed under field conditions and experimental results in the control of bindweed seedlings. Agron. J. 41: 130-133. Tuinstra, K. E. 1967. Vegetation of the floodplains and first terraces of Rock Creek near Red Lodge, Montana. Ph.D dissertation. Montana State University, Bozeman 110 pp.
Tuinstra, K. E. 1967. Vegetation of the floodplains and first terraces of Rock Creek near Red Lodge, Montana. Ph.D dissertation. Montana State University, Bozeman 110 pp. Waddington, K. D. 1976. Foraging patterns of halictid bees at flowers of Convolvulus arvensis L. Psyche 83: 112-119.
Waddington, K. D. 1976. Foraging patterns of halictid bees at flowers of Convolvulus arvensis L. Psyche 83: 112-119. Weaver, S. E., and W. R. Riley. 1982. The biology of Canadian weeds. 53. Convolvulus arvensis L. Can. J. Plant Sci. 62: 461-472.
Weaver, S. E., and W. R. Riley. 1982. The biology of Canadian weeds. 53. Convolvulus arvensis L. Can. J. Plant Sci. 62: 461-472. Whitesides, R. E. 1979. Field bindweed: a growth stage indexing system and its relation to control with glyphosate. Ph.D. Thesis, Oregon State University, Corvallis, Oregon.
Whitesides, R. E. 1979. Field bindweed: a growth stage indexing system and its relation to control with glyphosate. Ph.D. Thesis, Oregon State University, Corvallis, Oregon. Wiman, N.G. 2001. Dynamics of leafy spurge (Euphorbia esula L.) infested plant communities influenced by flea beetles in the Aphthona complex (Colepotera: Chrysomelidae). M.Sc. Thesis. Bozeman, MT: Montana State University. 148 p.
Wiman, N.G. 2001. Dynamics of leafy spurge (Euphorbia esula L.) infested plant communities influenced by flea beetles in the Aphthona complex (Colepotera: Chrysomelidae). M.Sc. Thesis. Bozeman, MT: Montana State University. 148 p. Wood, A.K. 1987. Ecology of a prairie mule deer population. Ph.D. Dissertation. Bozeman, MT: Montana State University. 205 p.
Wood, A.K. 1987. Ecology of a prairie mule deer population. Ph.D. Dissertation. Bozeman, MT: Montana State University. 205 p.
- Web Search Engines for Articles on "Field Bindweed"





