View in other NatureServe Network Field Guides
NatureServe
Montana
Utah
Wyoming
Idaho
Wisconsin
British Columbia
South Carolina
Yukon
California
New York
- Home - Other Field Guides
- Kingdom - - Animalia
- Phylum - Spiders, Insects, and Crustaceans - Arthropoda
- Class - Insects - Insecta
- Order - Sawflies / Wasps / Bees / Ants - Hymenoptera
- Family - Bumble, Honey, Carpenter, Stingless, & Orchid Bees - Apidae
- Species - Golden Northern Bumble Bee - Bombus fervidus
Golden Northern Bumble Bee - Bombus fervidus
Other Names:
Yellow Bumble Bee,
Thoracobombus fervidus, Bombus fervidus Complex
Native Species
Global Rank:
GNR
State Rank:
S5
(see State Rank Reason below)
Agency Status
USFWS:
USFS:
BLM:
State Rank Reason (see State Rank above)
Species is widely distributed across Montana and relatively common within suitable habitat. Trend appears stable and does not appear to face significant threats.
General Description
Koch et al. (2018) conducted morphometric and genetic analysis to examine the phylogenetic relationships of
Bombus fervidus and
Bombus californicus, both of which occur in Montana, and concurred with previous studies that individuals cannot be identified to species based on current taxonomy (i.e., setal color). However, Koch et al. (2018) found: (1) microsatellite genotypes have the capacity to identify two distinct genetic lineages in the
Bombus fervidus species complex in areas where the species are broadly sympatric and using the same habitats (including within Montana); (2) sympatric populations within the species complex appear to be phenotypically divergent and reproductively isolated with no evidence of recent introgression (diverging from a common ancestor likely less than ~1 million years ago); and (3) future research evaluating taxonomic characters like wing venation in conjunction with genetic analysis may identify morphological methods for distinguishing between these currently cryptic species.
For definitions and diagrams of bumble bee morphology please see the
Montana State Entomology Collection's Bumble Bee Morphology page. A long-tongued, medium-sized bumble bee: queens 18-21 mm in length, workers 11-17 mm. Head long, cheek distinctly longer than wide; mid-leg basitarsus with far back corner narrowly extended as a spine, outer surface of hind-leg tibia smooth and hairless (except fringe) forming pollen basket; hair on face black or predominantly so; upper side of thorax with black band between wings often narrow and sometimes intermixed with yellow hairs or lacking black hairs entirely, sides of thorax yellow at least in upper half; T1-4 predominantly yellow, T5 black. Males 13-16 mm in length; eyes similar in size and shape to eyes of any female bumble bee; antennae long, flagellum 4X the length of scape; hair color pattern similar to queens and workers, but hair of face and top of head black with minority of yellow intermixed (Colla et al. 2011, Koch et al. 2012, Williams et al. 2014).
Phenology
[Former B. californicus now considered this species.] Across the range, queens and workers reported March to October, males April to November (Williams et al. 2014). In Utah, queens reported April to June, workers April to September, males June to October (Koch et al. 2012); in southern Ontario, queens April to October, workers and males May to October (Colla and Dumesh 2010). In California (former B. californicus now considered this species), queens reported early February to early October, workers early March to late October, males early May to late October (Thorp et al. 1983).
Diagnostic Characteristics
Please see the
Montana State Entomology Collection's Key to Female Bumble Bees in Montana. [Former
B. californicus now considered this species.] Females told from other Montana
Bombus by a combination of the outer surface of hind-leg tibia concave and hairless (except fringe) forming pollen basket; cheek distinctly longer than wide; face predominantly with black hairs; hairs between wing bases completely or predominantly black; T1-4 predominantly yellow, T5-6 black.
Species Range
Montana Range
Range Descriptions
 Native
Native
Recorded Montana Distribution
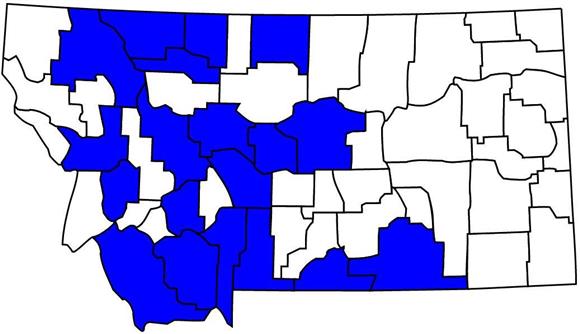
Click the map for additional distribution information.
Range Comments
[Former B. californicus now considered this species.] Widespread across southern Canada and the United States except the southern Great Plains and southeastern Gulf states (Colla et al. 2011, Koch et al. 2012, Williams et al. 2014). In California, coastal to at least 1525 m elevation (Thorp et al. 1983); in Colorado, 1600-2900 m elevation, but mostly below 2600 m (Macior 1974). Remains widespread in eastern North America but declining in abundance (Colla and Packer 2008, Grixti et al. 2009, Colla et al. 2012).
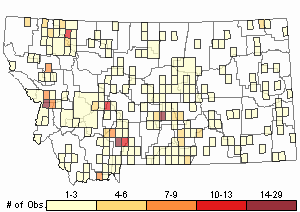
Observations in Montana Natural Heritage Program Database
Number of Observations: 462
(Click on the following maps and charts to see full sized version)
Map Help and Descriptions
Relative Density
Recency
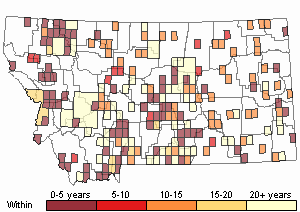
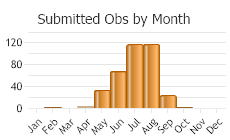
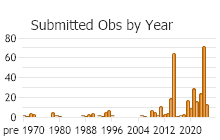
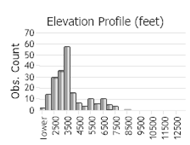 (Observations spanning multiple months or years are excluded from time charts)
(Observations spanning multiple months or years are excluded from time charts)
Habitat
[Former B. californicus now considered this species.] Open farmland and fields, tallgrass prairie, urban parks and gardens, urban prairie open spaces, sagebrush steppe, aspen parkland, montane meadows (Macior 1974, Richards 1978, Hines and Hendrix 2005, McFrederick and LeBuhn 2006, Wojcik et al. 2008, Colla and Dumesh 2010, Wilson et al. 2010, Cook et al. 2011, Molumby and Przybylowicz 2012). Also found infrequently in commercial Vaccinium cropland (Ratti et al. 2008).
Food Habits
[Former B. californicus now considered this species.] Feeds on a variety of flowers, including Ajuga, Anaphalis, Astragalus, Balsamorhiza, Calypso, Caragana, Carduus, Castilleja, Centaurea, Chrysothamnus, Cichorium, Cirsium, Cleome, Coronilla, Delphinium, Digitalis, Dipsacus, Echium, Epilobium, Erigeron, Frasera, Gentiana, Geranium, Grindelia, Helianthus, Hydrophyllum, Hypericum, Impatiens, Iris, Kalmia, Lactuca, Lathyrus, Linaria, Lobelia, Lonicera, Lotus, Lupinus, Lythrum, Malus, Medicago, Melilotus, Mentzelia, Monarda, Nepeta, Onobrychis, Onopordum, Oxytropis, Pedicularis, Penstemon, Phacelia, Potentilla, Prenanthes, Prunella, Rhus, Ribes, Robinia, Rosa, Rudbeckia, Salix, Sisyrinchium, Solanum, Solidago, Sonchus, Spiraea, Spiranthes, Stachys, Symphyotrichum, Symphytum, Taraxacum, Thermopsis, Trifolium, Vaccinium, Vicia and Wyethia (Plath 1934, Macior 1968, Beattie et al. 1973, Macior 1974, Heinrich 1976, Ackerman 1981, Thorp et al. 1983, Mayer et al. 2000, Ratti et al. 2008, Colla and Dumesh 2010, Wilson et al. 2010, Colla et al. 2011, Koch et al. 2012, Williams et al. 2014, Miller-Struttmann and Galen 2014, Ogilvie and Thomson 2015). Also rarely commercial Vaccinium (highbush blueberry) in southern British Columbia (Ratti et al. 2008).
Reproductive Characteristics
[Former B. californicus now considered this species.] Nests often built under ground, on the ground surface, or above ground in deserted rodent nests. Of 49 nests in the Boston area 61.2% were built underground, 32.6% were surface nests, and 6.1% were built above ground in stone walls or the eve of a shed (Plath 1934). Two of three Michigan nests were built in old mouse nests 61 and 183 cm above ground, the third nest was on the ground under a hay bale (Snider and Husband 1966). In southern Alberta, one study (Hobbs 1966a) reported 41.9% of 31 nests were built under ground and 58.1% on the surface. In a second Alberta study in aspen parkland (Richards 1978), 37.3% of 59 nests were built under ground, 33.9% on the ground surface, and 28.8% above the ground. Nests established in late May through June to mid July. Entrances to ground nests are frequently camouflaged; queens sometimes attempt to usurp nests from conspecifics (Richards 1978). First broods sometimes are small. A single egg is laid in each cell, the number of pupae (cocoons) in first broods averages 8-10. It takes an average of 26-28 days for workers to be produced from first broods. In second and third broods, 2-7 eggs are laid per cell (most often 3-4 eggs). Number of cocoons produced can vary widely, from 27 to 287. Workers of later broods become progressively larger, approaching queen size (Hobbs 1966a); workers have an adult life expectancy of 22-34 days (Goldblatt and Fell 1987). Males perch or hover outside nest entrances in search of queens. Queens dig hibernacula in the ground, to about 5 cm in captivity but probably deeper in the wild, and overwinter. This species parasitized by the cuckoo bumble bees Bombus insularis and B. suckleyi (Hobbs 1966a, Williams et al. 2014).
Stewardship Responsibility
References
- Literature Cited AboveLegend:
 View Online Publication
View Online Publication Ackerman, J.D. 1981. Pollination biology of Calypso bulbosa var. occidentalis (Orchidaceae): a food-deception system. Madroño 28(3): 101-110.
Ackerman, J.D. 1981. Pollination biology of Calypso bulbosa var. occidentalis (Orchidaceae): a food-deception system. Madroño 28(3): 101-110. Beattie, A.J., D.E. Breedlove, and P.R. Ehrlich. 1973. The ecology of the pollinators and predators of Frasera speciosa. Ecology 54: 81-91.
Beattie, A.J., D.E. Breedlove, and P.R. Ehrlich. 1973. The ecology of the pollinators and predators of Frasera speciosa. Ecology 54: 81-91. Colla, S., L. Richardson, and P. Williams. 2011. Bumble bees of the eastern United States. Washington, DC: USDA Forest Service, Pollinator Partnership. 103 p.
Colla, S., L. Richardson, and P. Williams. 2011. Bumble bees of the eastern United States. Washington, DC: USDA Forest Service, Pollinator Partnership. 103 p. Colla, S.R. and L. Packer. 2008. Evidence for decline in eastern North American bumble bees (Hymenoptera: Apidae), with special focus on Bombus affinis Cresson. Biodiversity Conservation 17: 1379-1391.
Colla, S.R. and L. Packer. 2008. Evidence for decline in eastern North American bumble bees (Hymenoptera: Apidae), with special focus on Bombus affinis Cresson. Biodiversity Conservation 17: 1379-1391. Colla, S.R. and S. Dumesh. 2010. The bumble bees of southern Ontario: notes on natural history and distribution. Journal of the Entomological Society of Ontario 141:39-68.
Colla, S.R. and S. Dumesh. 2010. The bumble bees of southern Ontario: notes on natural history and distribution. Journal of the Entomological Society of Ontario 141:39-68. Colla, S.R., F. Gadallah, L. Richarson, D. Wagner, and L. Gall. 2012. Assessing declines of North American bumble bees (Bombus spp.) using museum specimens. Biodiversity and Conservation 21: 3585-3595.
Colla, S.R., F. Gadallah, L. Richarson, D. Wagner, and L. Gall. 2012. Assessing declines of North American bumble bees (Bombus spp.) using museum specimens. Biodiversity and Conservation 21: 3585-3595. Cook, S.P., S.A. Birch, F.W. Merickel, C.C. Lowe, and D. Page-Dumroese. 2011. Bumble bee (Hymenoptera: Apidae) community structure on two sagebrush steppe sites in southern Idaho. Pan-Pacific Entomologist 87(3): 161-171.
Cook, S.P., S.A. Birch, F.W. Merickel, C.C. Lowe, and D. Page-Dumroese. 2011. Bumble bee (Hymenoptera: Apidae) community structure on two sagebrush steppe sites in southern Idaho. Pan-Pacific Entomologist 87(3): 161-171. Goldblatt, J.W. and R.D. Fell. 1987. Adult longevity of workers of the bumble bees Bombus fervidus (F.) and Bombus pennsylvanicus (De Geer)(Hymenoptera: Apidae). Canadian Journal of Zoology 65:2349-2353.
Goldblatt, J.W. and R.D. Fell. 1987. Adult longevity of workers of the bumble bees Bombus fervidus (F.) and Bombus pennsylvanicus (De Geer)(Hymenoptera: Apidae). Canadian Journal of Zoology 65:2349-2353. Grixti, J.C., L.T. Wong, S.A. Cameron, and C. Favret. 2009. Decline of bumble bees (Bombus) in the North American Midwest. Biological Conservation 142: 75-84.
Grixti, J.C., L.T. Wong, S.A. Cameron, and C. Favret. 2009. Decline of bumble bees (Bombus) in the North American Midwest. Biological Conservation 142: 75-84. Heinrich, B. 1976. Resource partitioning among some eusocial insects: bumblebees. Ecology 57(5): 874-889.
Heinrich, B. 1976. Resource partitioning among some eusocial insects: bumblebees. Ecology 57(5): 874-889. Hines, H.M. and S.D. Hendrix. 2005. Bumble bee (Hymenoptera: Apidae) diversity and abundance in tallgrass priaire patches: effects of local and landscape floral resources. Environmental Entomology 34(6): 1477-1484.
Hines, H.M. and S.D. Hendrix. 2005. Bumble bee (Hymenoptera: Apidae) diversity and abundance in tallgrass priaire patches: effects of local and landscape floral resources. Environmental Entomology 34(6): 1477-1484. Hobbs, G.A. 1966a. Ecology of species of Bombus Latr. (Hymenoptera: Apidae) in southern Alberta. IV. Subgenus Fervidobombus Skorikov. Canadian Entomologist 98: 33-39.
Hobbs, G.A. 1966a. Ecology of species of Bombus Latr. (Hymenoptera: Apidae) in southern Alberta. IV. Subgenus Fervidobombus Skorikov. Canadian Entomologist 98: 33-39. Koch, J., J. Strange, and P. Williams. 2012. Bumble bees of the western United States. Washington, DC: USDA Forest Service, Pollinator Partnership. 143 p.
Koch, J., J. Strange, and P. Williams. 2012. Bumble bees of the western United States. Washington, DC: USDA Forest Service, Pollinator Partnership. 143 p. Macior, L.M. 1968. Bombus (Hymenoptera, Apidae) queen foraging in relation to vernal pollination in Wisconsin. Ecology 49:20-25.
Macior, L.M. 1968. Bombus (Hymenoptera, Apidae) queen foraging in relation to vernal pollination in Wisconsin. Ecology 49:20-25. Macior, L.M. 1974. Pollination ecology of the Front Range of the Colorado Rocky Mountains. Melanderia 15: 1-59.
Macior, L.M. 1974. Pollination ecology of the Front Range of the Colorado Rocky Mountains. Melanderia 15: 1-59. Mayer, D.F., E.R. Miliczky, B.F. Finnigan, and C.A. Johnson. 2000. The bee fauna (Hymenoptera: Apoidea) of southeastern Washington. Journal of the Entomological Society of British Columbia 97: 25-31.
Mayer, D.F., E.R. Miliczky, B.F. Finnigan, and C.A. Johnson. 2000. The bee fauna (Hymenoptera: Apoidea) of southeastern Washington. Journal of the Entomological Society of British Columbia 97: 25-31. McFrederick, Q.S. and G. LeBuhn. 2006. Are urban parks refuges for bumble bees Bombus spp. (Hymenoptera: Apidae)? Biological Conservation 129: 372-382.
McFrederick, Q.S. and G. LeBuhn. 2006. Are urban parks refuges for bumble bees Bombus spp. (Hymenoptera: Apidae)? Biological Conservation 129: 372-382. Miller-Struttmann, N.E. and C. Galen. 2014. High-altitude multi-taskers: bumble bee food plant use broadens along an altitudinal productivity gradient. Oecologia 176:1033-1045.
Miller-Struttmann, N.E. and C. Galen. 2014. High-altitude multi-taskers: bumble bee food plant use broadens along an altitudinal productivity gradient. Oecologia 176:1033-1045. Molumby, A. and T. Przybylowicz. 2012. Bees (Hymenoptera: Apoidea) of the Chicago area: diversity and habitat use in an urbanized landscape. Great Lakes Entomologist 45:79-98.
Molumby, A. and T. Przybylowicz. 2012. Bees (Hymenoptera: Apoidea) of the Chicago area: diversity and habitat use in an urbanized landscape. Great Lakes Entomologist 45:79-98. Ogilvie, J.E. and J.D. Thomson. 2015. Male bumble bees are important pollinators of a late-blooming plant. Arthropod-Plant Interactions 9:205-213.
Ogilvie, J.E. and J.D. Thomson. 2015. Male bumble bees are important pollinators of a late-blooming plant. Arthropod-Plant Interactions 9:205-213. Plath, O.E. 1934. Bumblebees and their ways. New York, NY: Macmillan Company. 201 p.
Plath, O.E. 1934. Bumblebees and their ways. New York, NY: Macmillan Company. 201 p. Ratti, C.M., H.A. Higo, T.L. Griswold, and M.L. Winston. 2008. Bumble bees influence berry size in comercial Vaccinium spp. cultivation in British Columbia. Canadian Entomologist 140(3): 348-363.
Ratti, C.M., H.A. Higo, T.L. Griswold, and M.L. Winston. 2008. Bumble bees influence berry size in comercial Vaccinium spp. cultivation in British Columbia. Canadian Entomologist 140(3): 348-363. Richards, K.W. 1978. Nest site selection by bumble bees (Hymenoptera: Apidae) in southern Alberta. Canadian Entomologist 110(3): 301-318.
Richards, K.W. 1978. Nest site selection by bumble bees (Hymenoptera: Apidae) in southern Alberta. Canadian Entomologist 110(3): 301-318. Snider, R.J. and R.W. Husband. 1966. Collembola found in bumblebee nests. Transactions of the American Microscopical Society 85:473-475.
Snider, R.J. and R.W. Husband. 1966. Collembola found in bumblebee nests. Transactions of the American Microscopical Society 85:473-475. Thorp, R.W., D.S. Horning, and L.L. Dunning. 1983. Bumble bees and cuckoo bumble bees of California (Hymenoptera: Apidae). Bulletin of the California Insect Survey 23:1-79.
Thorp, R.W., D.S. Horning, and L.L. Dunning. 1983. Bumble bees and cuckoo bumble bees of California (Hymenoptera: Apidae). Bulletin of the California Insect Survey 23:1-79. Williams, P., R. Thorp, L. Richardson, and S. Colla. 2014. Bumble Bees of North America. Princeton, NJ: Princeton University Press. 208 p.
Williams, P., R. Thorp, L. Richardson, and S. Colla. 2014. Bumble Bees of North America. Princeton, NJ: Princeton University Press. 208 p. Wilson, J.S., L.E. Wilson, L.D. Loftis, and T. Griswold. 2010. The montane bee fauna of north central Washington, USA, with floral associations. Western North American Naturalist 70(2): 198-207.
Wilson, J.S., L.E. Wilson, L.D. Loftis, and T. Griswold. 2010. The montane bee fauna of north central Washington, USA, with floral associations. Western North American Naturalist 70(2): 198-207. Wojcik, V.A., G.W. Frankie, R.W. Thorp, and J.L. Hernandez. 2008. Seasonality in bees and their floral resource plants at a constructed urban bee habitat in Berkeley, California. Journal of the Kansas Entomological Society 81: 15-28.
Wojcik, V.A., G.W. Frankie, R.W. Thorp, and J.L. Hernandez. 2008. Seasonality in bees and their floral resource plants at a constructed urban bee habitat in Berkeley, California. Journal of the Kansas Entomological Society 81: 15-28.
- Additional ReferencesLegend:
 View Online Publication
View Online Publication
Do you know of a citation we're missing? Adhikari, S. 2018. Impacts of dryland farming systems on biodiversity, plant-insect interactions, and ecosystem services. Ph.D. Dissertation. Bozeman, MT: Montana State University. 207 p.
Adhikari, S. 2018. Impacts of dryland farming systems on biodiversity, plant-insect interactions, and ecosystem services. Ph.D. Dissertation. Bozeman, MT: Montana State University. 207 p. Burkle L.A., M.P. Simanonok, J.S. Durney, J.A. Myers, and R.T. Belote. 2019. Wildfires influence abundance, diversity, and intraspecific and interspecific trait variation of native bees and flowering plants across burned and unburned landscapes. Frontiers in Ecology and Evolution 7(252):1-14.
Burkle L.A., M.P. Simanonok, J.S. Durney, J.A. Myers, and R.T. Belote. 2019. Wildfires influence abundance, diversity, and intraspecific and interspecific trait variation of native bees and flowering plants across burned and unburned landscapes. Frontiers in Ecology and Evolution 7(252):1-14. Delphia, C.M., Griswold, T., Reese, E.G., O'Neill, K.M., and Burkle, L.A. 2019. Checklist of bees (Hymenoptera: Apoidea) from small, diversified vegetable farms in south-western Montana. Biodiversity Data Journal: e30062
Delphia, C.M., Griswold, T., Reese, E.G., O'Neill, K.M., and Burkle, L.A. 2019. Checklist of bees (Hymenoptera: Apoidea) from small, diversified vegetable farms in south-western Montana. Biodiversity Data Journal: e30062 Dolan, A.C. 2016. Insects associated with Montana's huckleberry (Ericaceae: Vaccinium globulare) plants and the bumble bees (Hymenoptera: Apidae) of Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 160 p.
Dolan, A.C. 2016. Insects associated with Montana's huckleberry (Ericaceae: Vaccinium globulare) plants and the bumble bees (Hymenoptera: Apidae) of Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 160 p. Dolan, A.C., C.M. Delphia, K.M. O'Neill, and M.A. Ivie. 2017. Bumble Bees (Hymenoptera: Apidae) of Montana. Annals of the Entomological Society of America. 110(2): 129-144.
Dolan, A.C., C.M. Delphia, K.M. O'Neill, and M.A. Ivie. 2017. Bumble Bees (Hymenoptera: Apidae) of Montana. Annals of the Entomological Society of America. 110(2): 129-144. Fultz, J.E. 2005. Effects of shelterwood management on flower-visiting insects and their floral resources. M.Sc. Thesis. Bozeman, MT: Montana State University. 163 p.
Fultz, J.E. 2005. Effects of shelterwood management on flower-visiting insects and their floral resources. M.Sc. Thesis. Bozeman, MT: Montana State University. 163 p. Kearns, C.A. and J.D. Thomson. 2001. The Natural History of Bumble Bees. Boulder, CO. University Press of Colorado.
Kearns, C.A. and J.D. Thomson. 2001. The Natural History of Bumble Bees. Boulder, CO. University Press of Colorado. Reese, E.G., L.A. Burkle, C.M. Delphia, and T. Griswold. 2018. A list of bees from three locations in the Northern Rockies Ecoregion (NRE) of western Montana. Biodiversity Data Journal 6: e27161.
Reese, E.G., L.A. Burkle, C.M. Delphia, and T. Griswold. 2018. A list of bees from three locations in the Northern Rockies Ecoregion (NRE) of western Montana. Biodiversity Data Journal 6: e27161. Sater, S. 2022. The insects of Sevenmile Creek, a pictorial guide to their diversity and ecology. Undergraduate Thesis. Helena, MT: Carroll College. 242 p.
Sater, S. 2022. The insects of Sevenmile Creek, a pictorial guide to their diversity and ecology. Undergraduate Thesis. Helena, MT: Carroll College. 242 p. Simanonok, M. 2018. Plant-pollinator network assembly after wildfire. Ph.D. Dissertation. Bozeman, MT: Montana State University. 123 p.
Simanonok, M. 2018. Plant-pollinator network assembly after wildfire. Ph.D. Dissertation. Bozeman, MT: Montana State University. 123 p. Simanonok, M.P. and L.A. Burkle. 2019. Nesting success of wood-cavity-nesting bees declines with increasing time since wildfire. Ecology and Evolution 9:12436-12445.
Simanonok, M.P. and L.A. Burkle. 2019. Nesting success of wood-cavity-nesting bees declines with increasing time since wildfire. Ecology and Evolution 9:12436-12445.
- Web Search Engines for Articles on "Golden Northern Bumble Bee"
- Additional Sources of Information Related to "Insects"





