View in other NatureServe Network Field Guides
NatureServe
Montana
Utah
Wyoming
Idaho
Wisconsin
British Columbia
South Carolina
Yukon
California
New York
Lesser Bladderwort - Utricularia minor
General Description
PLANTS: Perennial aquatic herbs with submerged stems, bladders, and overwintering buds. Plants are rootless with slender stems. Bladders, which trap algae and small aquatic animals for the plants nourishment, are borne on leafy, submerged branches. Source: Lesica et al. 2012.
LEAVES: The leaves are mostly 2-8 mm long, palmately divided, with flat and entire segments. Source: Lesica et al. 2012.
INFLORESCENCE: An erect, few flowered, bracteate raceme. 2 to 5 flowers are emergent, yellow, and snapdragon-like. Source: Lesica et al. 2012.
The specific epithet minor refers to this species’ smaller size compared to others within the genus. Utricularia is derived from the Latin utriculus which translates to wineskin or leather bottle referring to the sac-like bladders of the genus (Poppinga et al. 2016).
Phenology
Flowering May through September (Neid 2006).
Diagnostic Characteristics
Montana has four
Utricularia species. They are all aquatic, submerged, rootless herbs with small bladder-like traps borne on branches or leaves. All species have emergent, snapdragon-like flowers borne on a scape. All have many, globose overwintering buds called turions.
Lesser Bladderwort –
Utricularia minor, native and desirable
*Size: Scape emerges 5-10 cm from the water.
*Bladders: 1-3 mm long and occur only on leafy stems.
*Bladder Glands: Long arm-pair diverging slightly. Short arm-pair diverging by 270–300 degrees (so all 4 arms oriented in the same direction) (Rice 2012).
*Leaves: 2-8 mm long and palmately divided. Segments are flat with entire margins.
*Buds: 2-9 mm long.
*Flowers: 2-5 yellow flowers in an inflorescence. Each is 5-10 mm long with a 2 mm long spur.
*Fruit: Borne on curved pedicels when mature.
Flatleaf Bladderwort –
Utricularia intermedia, native, SOC
*Size: Scape emerges 8-25 cm from the water.
*Bladders: 3-5 mm long and borne on separate leafless branches.
*Bladder Glands: Both arm-pairs generally diverge by 0–30 degrees (Rice 2012).
*Leaves: 5-20 mm long, palmately divided. Segments are flat with entire to obscurely serrate margins.
*Buds: 6-15 mm long.
*Flowers: 1-4 yellow flowers in the inflorescence. Each is 10-15 mm long with a 5-9 mm long spur.
*Fruit: Borne on erect pedicels when mature.
Greater Bladderwort –
Utricularia vulgaris, native and desirable
*Size: Scape emerges 10-40 cm from the water.
*Bladders: 2-5 mm long and only attached to leaf segments.
*Bladder Glands: Long arm-pair is parallel or diverges slightly. Short arm-pair diverges by 90–180 degrees (Rice 2012).
*Leaves: Pinnately divided and crowded. 20-50 mm long, entire and more or less round.
*Buds: 15-25 mm long.
*Flowers: More than 5 yellow flowers in the inflorescence. Each is 10-18 mm long with a 5-8 mm long spur.
*Fruit: Borne on curved pedicels when mature.
Northern Bladderwort –
Utricularia ochroleuca, native, SOC
*Size: Scape emerges up to 15 cm from the water
*Bladders: Most are borne on leafless stems but a few can often be found on leafy shoots.
*Bladder Glands: long arm-pair diverging by 20–45°, short arm-pair diverging by 40–160 degrees (Rice 2012)
*Leaves: 5-15 mm long and palmately divided. Terminal leaf segments with bristle tips and toothed margins.
*Buds: 2-3 mm and bristly
*Flowers: Inflorescence usually 3-5 flowered. Each is 8-11 mm long with a 10-15 mm long spur.
*Fruit: Borne on curved pedicels when mature.
Species Range
Montana Range
Range Descriptions

 Native
Native
Range Comments
Circumboreal south to CA, CO, IA and NJ (Lesica et al. 2012. Manual of Montana Vascular Plants. BRIT Press. Fort Worth, TX).
Observations in Montana Natural Heritage Program Database
Number of Observations: 77
(Click on the following maps and charts to see full sized version)
Map Help and Descriptions
Relative Density
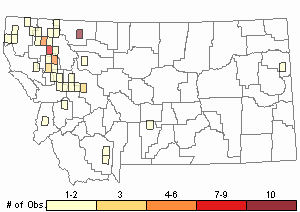
Recency
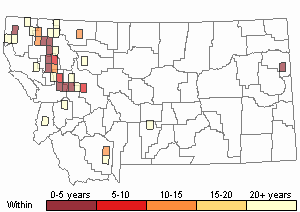
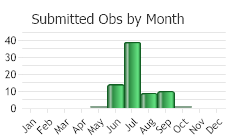
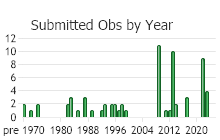
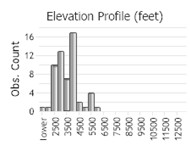 (Observations spanning multiple months or years are excluded from time charts)
(Observations spanning multiple months or years are excluded from time charts)
Habitat
Ecology
Utricularia ochroleuca is most likely a hybrid of U. minor x U. intermedia (Rice 2012).
BLADDER FUNCTION
All Bladderwort species are carnivorous plants that use small bladder-like traps to capture and digest prey. Prey includes aquatic insect larvae, water mites, nematodes, gastropods, small tadpoles, crustaceans, diatoms, and other aquatic microorganisms (Mette et al. 2000). Each trap has a valve that opens like a trap door when the surrounding trigger hairs are brushed (Poppinga et al. 2016). When the valve opens, a low pressure gradient causes water to flow quickly inside pulling in the organism that triggered the hairs (Singh et al. 2011). Glands found on the inside of the traps both digest prey through enzyme excretion, and absorb the nutrients after digestion (Poppinga et al. 2016).
UTRICULARIA AND ALGAE
While Utricularia are considered “carnivorous” plants, their nutrient acquisition is much more complicated than carnivory alone. In fact, algae have been observed inside bladders more often than anything else (Plachno et al. 2012). This has caused a close and complex relationship to develop between Utricularia species and algae. Individuals trap and digest algae for nutrients, and also host algae and other microorganisms that live within traps, creating an elaborate food-web community (Peroutka et al. 2008; Ellwood et al. 2018). Fungi, bacteria, and protozoa are cultivated within traps and help to break down algae into a state that can be digested by the bladderwort (Sirová et al. 2018).
According to a study by Ulanowicz (1995), Utricularia species use a positive feedback loop in order to survive in an oligotrophic environment in which other aquatic plant species would be nutrient stressed. By acting as a physical structure for periphyton to grow on, they attract the organisms that graze on periphyton which they will then feed upon (Ulanowicz 1995).
PHOTOSYNTHESIS AND RESPIRATION
In addition to preying on a wide variety of other organisms, bladderworts photosynthesize in order to obtain energy. Adamec (2005) showed that the photosynthetic rate of leaves was much higher than that of bladders whereas respiration of bladders was much higher than that of the leaves. This implies that while bladders obtain essential nitrogen and phosphorous from digesting prey, function of these structures necessitates high metabolic costs (Adamec 2005). While the benefits of carnivory in Utricularia are not fully understood, this investment of energy suggests some net advantage to trap operation.</u.></u.>
Reproductive Characteristics
FLOWERS [Lesica et al. 2012; Hitchcock and Cronquist 2018]
Emergent flowers have an inconspicuous, two-lobed calyx and a dull yellow, two-lipped corolla. The corolla is usually 5-10 mm long. Underneath, it has a spur, which is about 2 mm long; above, it has a small to obsolete palate. The upper corolla lip is less than half the length of the lower lip.
FRUIT
The fruit is a circumscissile capsule 1 mm long or less that is borne on a curved pedicel (Lesica et al. 2012; Rice 2012)
LIFE CYCLE [Adamec 1999]
U. minor is a perennial species. Its turions begin forming in late August and are surrounded by a matrix of branched leaves. By late September and early October, branches die and drag the turions to the bottom. In late autumn, the turions break free but remain submerged and tangled in the branches. Once the branches fully decompose, the turions float to the surface and begin new growth in the spring.
Management
Conservation impacts on Utricularia minor are essentially unstudied, but populations are known to be particularly sensitive to changes in water quality. Efforts to maintain and improve water quality and preserve suitable habitat should therefore be taken to sustain populations. This may include monitoring hydrologic impacts on watersheds where Utricularia minor is found, and establishing buffers around identified locations. Source: Neid 2006.
As nutrient levels rise in oligotrophic habitats, periphyton and algae growth will begin to choke-out Utricularia plants, at which point they no longer benefit from attracting algae (Ulanowicz 1994). In this situation, algae-suppressive treatments are harmful to bladderworts that are caught amongst the algae. Introducing Daphnia and tadpoles is the only known method of suppressing algae that does not harm Utricularia. Source: Carnivorous Plant Resource.
Stewardship Responsibility
Threats or Limiting Factors
Invasive species, habitat loss and hydrologic alterations are the main threats to Utricularia minor. This species is very sensitive to changes in water quality so land use practices that have a high impact on both local and landscape scale habitat integrity also threaten populations. Furthermore, fens, which are the primary habitat of Lesser Bladderwort, are both restricted in range and sensitive to environmental change. Source: Neid 2006.
While a certain level of nutrients can attract a beneficial amount of algae and periphyton, very high nutrient levels resulting in unchecked growth of algae can displace Utricularia populations (Ulanowicz 1994).
References
- Literature Cited AboveLegend:
 View Online Publication
View Online Publication Adamec, L. 2005. Respiration and Photosynthesis of Bladders and Leaves of Aquatic Utricularia Species. Plant Biology 8: 765-769.
Adamec, L. 2005. Respiration and Photosynthesis of Bladders and Leaves of Aquatic Utricularia Species. Plant Biology 8: 765-769. Adamec, Lubomir. 1999. Turion overwintering of aquatic carnivorous plants. Carnivorous Plant Newsletter 28: 19-24.
Adamec, Lubomir. 1999. Turion overwintering of aquatic carnivorous plants. Carnivorous Plant Newsletter 28: 19-24. Barry A. Rice. 2012. Utricularia, in Jepson Flora Project (eds.) Jepson eFlora, /eflora/eflora_display.php?tid=10569, accessed on 7 November 2019
Barry A. Rice. 2012. Utricularia, in Jepson Flora Project (eds.) Jepson eFlora, /eflora/eflora_display.php?tid=10569, accessed on 7 November 2019 Ellwood, N.T.W., Congestri, R., and Ceschin, S. 2019. The role of phytoplankton in the diet of the bladderwort Utricularia australis R.Br. (Lentibulariaceae). Freshwater Biology 64: 233– 243.
Ellwood, N.T.W., Congestri, R., and Ceschin, S. 2019. The role of phytoplankton in the diet of the bladderwort Utricularia australis R.Br. (Lentibulariaceae). Freshwater Biology 64: 233– 243. Hitchcock, C.L. and A. Cronquist. 2018. Flora of the Pacific Northwest: An Illustrated Manual. Second Edition. Giblin, D.E., B.S. Legler, P.F. Zika, and R.G. Olmstead (eds). Seattle, WA: University of Washington Press in Association with Burke Museum of Natural History and Culture. 882 p.
Hitchcock, C.L. and A. Cronquist. 2018. Flora of the Pacific Northwest: An Illustrated Manual. Second Edition. Giblin, D.E., B.S. Legler, P.F. Zika, and R.G. Olmstead (eds). Seattle, WA: University of Washington Press in Association with Burke Museum of Natural History and Culture. 882 p. Lesica, P., M.T. Lavin, and P.F. Stickney. 2012. Manual of Montana Vascular Plants. Fort Worth, TX: BRIT Press. viii + 771 p.
Lesica, P., M.T. Lavin, and P.F. Stickney. 2012. Manual of Montana Vascular Plants. Fort Worth, TX: BRIT Press. viii + 771 p. Mette, N., N. Wilbert, and W. Barthlott. 2000. Food Composition of Aquatic Bladderworts (Utricularia, Lentibulariaceae) in Various Habitats. Beitr. Biol. Pflanzen 72: 1-13.
Mette, N., N. Wilbert, and W. Barthlott. 2000. Food Composition of Aquatic Bladderworts (Utricularia, Lentibulariaceae) in Various Habitats. Beitr. Biol. Pflanzen 72: 1-13. Neid, S.L. (2006, May 15). Utricularia minor L. (lesser bladderwort): a technical conservation assessment. [Online]. USDA Forest Service, Rocky Mountain Region. Available: https://www.fs.usda.gov/Internet/FSE_DOCUMENTS/stelprdb5206905.pdf [2019, November 7].
Neid, S.L. (2006, May 15). Utricularia minor L. (lesser bladderwort): a technical conservation assessment. [Online]. USDA Forest Service, Rocky Mountain Region. Available: https://www.fs.usda.gov/Internet/FSE_DOCUMENTS/stelprdb5206905.pdf [2019, November 7]. Peroutka, M., W. Adlassnig, M. Volgger, T. Lendl, W. Url, and I. Lichtscheidl. 2008. Utricularia: a vegetarian carnivorous plant? Plant Ecology 199: 153.
Peroutka, M., W. Adlassnig, M. Volgger, T. Lendl, W. Url, and I. Lichtscheidl. 2008. Utricularia: a vegetarian carnivorous plant? Plant Ecology 199: 153. Plachno B., Lukaszek M., Wolowski K., Adamec L., and Stolarczyk P. 2012. Aging of Utricularia traps and variability of microorganisms associated with that micro-habitat. Aquat Bot. 97:44–48.
Plachno B., Lukaszek M., Wolowski K., Adamec L., and Stolarczyk P. 2012. Aging of Utricularia traps and variability of microorganisms associated with that micro-habitat. Aquat Bot. 97:44–48. Poppinga, S., C. Weisskopf, A.S.Westermeier, T. Masselter, and T. Speck. 2016. Fastest predators in the plant kingdom: functional morphology and biomechanics of suction traps found in the largest genus of carnivorous plants. AoB PLANTS 8: 140. https://doi.org/10.1093/aobpla/plv140
Poppinga, S., C. Weisskopf, A.S.Westermeier, T. Masselter, and T. Speck. 2016. Fastest predators in the plant kingdom: functional morphology and biomechanics of suction traps found in the largest genus of carnivorous plants. AoB PLANTS 8: 140. https://doi.org/10.1093/aobpla/plv140 Singh, A.K., Prabhakar, S., and Sane, S.P. 2011. The biomechanics of fast prey capture in aquatic bladderworts. Biology Letters 7(4), 547–550
Singh, A.K., Prabhakar, S., and Sane, S.P. 2011. The biomechanics of fast prey capture in aquatic bladderworts. Biology Letters 7(4), 547–550 Sirova, D., J. Barta, K. Simek, T. Posch, J. Pech, J. Stone, J. Borovec, L. Adamec, and J. Vrba. 2018. Hunters or farmers? Microbiome characteristics help elucidate the diet composition in an aquatic carnivorous plant. Microbiome 6: 225.
Sirova, D., J. Barta, K. Simek, T. Posch, J. Pech, J. Stone, J. Borovec, L. Adamec, and J. Vrba. 2018. Hunters or farmers? Microbiome characteristics help elucidate the diet composition in an aquatic carnivorous plant. Microbiome 6: 225. Ulanowicz, R.E. 1995. Utricularia's secret: the advantage of positive feedback in oligotrophic environments. Ecological Modelling 79: 49-57.
Ulanowicz, R.E. 1995. Utricularia's secret: the advantage of positive feedback in oligotrophic environments. Ecological Modelling 79: 49-57.
- Additional ReferencesLegend:
 View Online Publication
View Online Publication
Do you know of a citation we're missing? Camp, P. and John G. Gamon. 2011. Field Guide to the Rare Plants of Washington. University of Washington Press.
Camp, P. and John G. Gamon. 2011. Field Guide to the Rare Plants of Washington. University of Washington Press. Koller-Peroutka, M., T. Lendl, M. Watzka, and W. Adlassnig. 2015. Capture of algae promotes growth and propagation in aquatic Utricularia. Annals of Botany 115(2): 227–236.
Koller-Peroutka, M., T. Lendl, M. Watzka, and W. Adlassnig. 2015. Capture of algae promotes growth and propagation in aquatic Utricularia. Annals of Botany 115(2): 227–236. Lesica, P., M.T. Lavin, and P.F. Stickney. 2022. Manual of Montana Vascular Plants, Second Edition. Fort Worth, TX: BRIT Press. viii + 779 p.
Lesica, P., M.T. Lavin, and P.F. Stickney. 2022. Manual of Montana Vascular Plants, Second Edition. Fort Worth, TX: BRIT Press. viii + 779 p. Maine Volunteer Lake Monitoring Program. 2009. Bladderworts. Maine's Interactive Field Guide to Aquatic Invaders (and Their Native Look Alikes). https://lakestewardsofmaine.org/mciap/herbarium/Bladderworts.php
Maine Volunteer Lake Monitoring Program. 2009. Bladderworts. Maine's Interactive Field Guide to Aquatic Invaders (and Their Native Look Alikes). https://lakestewardsofmaine.org/mciap/herbarium/Bladderworts.php
- Web Search Engines for Articles on "Lesser Bladderwort"





