View in other NatureServe Network Field Guides
NatureServe
Montana
Utah
Wyoming
Idaho
Wisconsin
British Columbia
South Carolina
Yukon
California
New York
European Honey Bee - Apis mellifera
General Description
Medium-sized, and fairly slow-flying relative to other bees in the Family: queens 17-18 mm in length, workers 11-12 mm, drones (males) 15-16 mm. Females (workers and to a much lesser extent queens) most often encountered, males (drones) rarely so; females with 10 flagellomeres (segments) in the flagellum of the antennae, males with 11 flagellomeres. Moderately hairy, especially on head and thorax; integument color highly variable from light orange to almost black, but usually with amber or yellowish areas on metasoma; hair present on compund eyes; mandibles lack teeth and have a spatulate, twisted appearance; hindleg tibial spurs absent, hindleg tibia and femur of workers broad, slightly concave, shiny and hairless (except fringe) forming a pollen basket (corbicula); hind legs often hang below body while in flight. Wings unique in North American bees north of Mexico; the marginal cell extremely long and extending almost to wing tip; stigma very small, the three submarginal cells are non-uniform in shape (Michener et al. 1994, Wilson and Carril 2016).
Phenology
In our region, active March to September with peak activity May to July. Active season longer or shorter elsewhere in North America, with some active somewhere in North America every month of the year (Wilson and Carrill 2016).
Diagnostic Characteristics
A combination of fluffy hairs, presence of pollen basket (corbicula) on hindleg tibia of workers, lack of hindleg tibial spurs, twisted and spatulate mandibles, hairs in compound eyes, and wings with extremely long marginal cell extending nearly to wing tip, a very small stigma, and 3 non-uniform submarginal cells, distinguish Apis from other North and Central American bee genera (Michener et al. 1994, Wilson and Carril 2016).
Species Range
Montana Range
Range Descriptions
 Non-native
Non-native
Range Comments
Worldwide except in Antarctica, above the Arctic Circle, and in the Sahara Desert; introduced in many regions (Wilson and Carril 2016). Widespread in Montana as a result of commercial use for crop pollination and honey production. Feral colonies establish in the wild from escaped swarms (Visscher and Seeley 1982, 1989; Seeley 1983, 1985), but published descriptions of feral colony locations are lacking for Montana. Elevation range not reported, but likely most abundant across the state at low elevations.
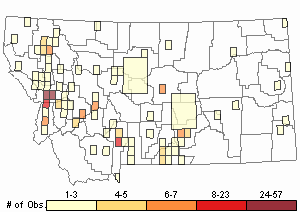
Observations in Montana Natural Heritage Program Database
Number of Observations: 500
(Click on the following maps and charts to see full sized version)
Map Help and Descriptions
Relative Density
Recency
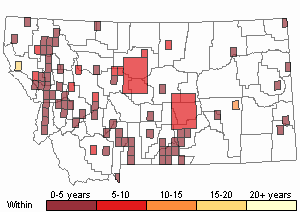
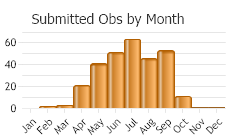
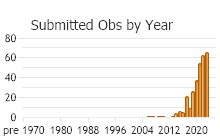
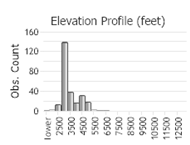 (Observations spanning multiple months or years are excluded from time charts)
(Observations spanning multiple months or years are excluded from time charts)
Habitat
Occurs in many habitats, including urban gardens and parks, railroad margins, prairie, grasslands, shrubland, fields, farmland, oak and mixed deciduous forests, dunes, semi-deserts, wetlands, riparian woodlands, and montane meadows (Visscher and Seeley 1982, Ratnieks et al. 1991, Baum et al. 2005, Wojcik et al. 2008, Molumby and Przybylowicz 2012). Colonies in artifical beehives occupy many kinds of commercial croplands (MacKenzie and Averill 1995, MacKenzie and Eickwort 1996, Calderone 2012).
Food Habits
Generalist, collecting pollen and nectar from nearly every type of flower, both native and domesticated (Wilson and Carril 2016).
Reproductive Characteristics
Feral colonies nest usually above ground to at least 8 m height, but sometimes at ground level. Colonies use a diversity of cavities for nest sites including tree hollows, arboreal termite nests, caves and rock crevices, abandoned buildings, barrels, bird boxes, and rarely in unprotected locations on tree branches; commercial colonies established and maintained in artificial beehives (Seeley and Morse 1976, 1978; Seeley 1983, 2010; Ratnieks et al. 1991; Baum et al. 2005; Wilson and Carril 2016). Queens establish new colonies after mating with 12-15 drones and storing sperm for the remainder of their lives (queens may live 2-7 years). At peak egg production, queens lay 1500-2000 eggs per day, and about 150,000 in a summer. Eggs hatch in 3 days. Duration of larval and pupal stages depends on caste. Larval stage is 6 days for workers, 6.5 days for drones, 5.5 days for queens; pupal stage is 12 days for workers, 14.5 days for drones, 8 days for queens. Swarming by old queens and workers starts in early summer following production of 10-15 new queens (Seeley 2010, Wilson and Carril 2016). Workers survive only a few months, drones about 3 months; foraging workers tend to die before reaching senescence, with a median lifespan of 7 days (range = 2-17 days). In some colonies, a very small fraction of drones are produced from eggs laid by workers (Visscher 1989, Visscher and Dukas 1997, Wilson and Carril 2016).
Management
Stewardship Responsibility
References
- Literature Cited AboveLegend:
 View Online Publication
View Online Publication Baum, K.A., W.L. Rubink, M.A. Pinto, and R.N. Coulson. 2005. Spatial and temporal distribution and nest site characteristics of feral honey bee (Hymenoptera: Apidae) colonies in a coastal prairie landscape. Environmental Entomology 34:610-618.
Baum, K.A., W.L. Rubink, M.A. Pinto, and R.N. Coulson. 2005. Spatial and temporal distribution and nest site characteristics of feral honey bee (Hymenoptera: Apidae) colonies in a coastal prairie landscape. Environmental Entomology 34:610-618. Calderone, N.W. 2012. Insect pollinated crops, insect pollinators and US agriculture: trend analysis of aggregated data for the period 1992-2009. PLoS ONE 7: e37235.
Calderone, N.W. 2012. Insect pollinated crops, insect pollinators and US agriculture: trend analysis of aggregated data for the period 1992-2009. PLoS ONE 7: e37235. MacKenzie, K.E. and A. L. Averill. 1995. Bee (Hymenoptera: Apoidea) diversity and abundance on cranberry in southeastern Massachusetts. Annals of the Entomological Society of America 88(3): 334-341.
MacKenzie, K.E. and A. L. Averill. 1995. Bee (Hymenoptera: Apoidea) diversity and abundance on cranberry in southeastern Massachusetts. Annals of the Entomological Society of America 88(3): 334-341. MacKenzie, K.E. and G.C. Eickwort. 1996. Diversity and abundance of bees (Hymenoptera: Apoidea) foraging on highbush blueberry (Vaccinium corymbosum L.) in central New York. Journal of the Kansas Entomological Society 69(4) (supplement): 185-194.
MacKenzie, K.E. and G.C. Eickwort. 1996. Diversity and abundance of bees (Hymenoptera: Apoidea) foraging on highbush blueberry (Vaccinium corymbosum L.) in central New York. Journal of the Kansas Entomological Society 69(4) (supplement): 185-194. Michener, C.D., R.J. McGinley, and B.N. Danforth. 1994. The bee genera of North and Central America. Washington, DC: Smithsonian Institution Press. 209 p.
Michener, C.D., R.J. McGinley, and B.N. Danforth. 1994. The bee genera of North and Central America. Washington, DC: Smithsonian Institution Press. 209 p. Molumby, A. and T. Przybylowicz. 2012. Bees (Hymenoptera: Apoidea) of the Chicago area: diversity and habitat use in an urbanized landscape. Great Lakes Entomologist 45:79-98.
Molumby, A. and T. Przybylowicz. 2012. Bees (Hymenoptera: Apoidea) of the Chicago area: diversity and habitat use in an urbanized landscape. Great Lakes Entomologist 45:79-98. Ratnieks, F.L.W., M.A. Piery, and I. Cuadriello. 1991. The natural nest and nest density of the Africanized honey bee (Hymenoptera, Apidae) near Tapachula, Chiapas, Mexico. Canadian Entomologist 123:353-359.
Ratnieks, F.L.W., M.A. Piery, and I. Cuadriello. 1991. The natural nest and nest density of the Africanized honey bee (Hymenoptera, Apidae) near Tapachula, Chiapas, Mexico. Canadian Entomologist 123:353-359. Seeley, T.D. 1983. The ecology of temperate and tropical honeybee societies. American Scientist 71:264-272.
Seeley, T.D. 1983. The ecology of temperate and tropical honeybee societies. American Scientist 71:264-272. Seeley, T.D. 1985. Honeybee ecology: a study of adaptation in social life. Princeton, NJ: Princeton University Press. 201 p.
Seeley, T.D. 1985. Honeybee ecology: a study of adaptation in social life. Princeton, NJ: Princeton University Press. 201 p. Seeley, T.D. 2010. Honeybee democracy. Princeton, NJ: Princeton University Press. 273 p.
Seeley, T.D. 2010. Honeybee democracy. Princeton, NJ: Princeton University Press. 273 p. Seeley, T.D. and R.A, Morse. 1976. The nest of the honey bee (Apis mellifera L.). Insectes Sociaux 23:495-512.
Seeley, T.D. and R.A, Morse. 1976. The nest of the honey bee (Apis mellifera L.). Insectes Sociaux 23:495-512. Seeley, T.D. and R.A. Morse. 1978. Nest site selection by the honey bee, Apis mellifera. Insectes Sociaux 25:323-337.
Seeley, T.D. and R.A. Morse. 1978. Nest site selection by the honey bee, Apis mellifera. Insectes Sociaux 25:323-337. Visscher, P.K. and R. Dukas. 1997. Survivorship of foraging honey bees. Insectes Sociaux 44:1-5.
Visscher, P.K. and R. Dukas. 1997. Survivorship of foraging honey bees. Insectes Sociaux 44:1-5. Visscher, P.K. and T.D. Seeley. 1982. Foraging strategy of honeybee colonies in a temperate deciduous forest. Ecology 63:1790-1801.
Visscher, P.K. and T.D. Seeley. 1982. Foraging strategy of honeybee colonies in a temperate deciduous forest. Ecology 63:1790-1801. Williams, P., R. Thorp, L. Richardson, and S. Colla. 2014. Bumble Bees of North America. Princeton, NJ: Princeton University Press. 208 p.
Williams, P., R. Thorp, L. Richardson, and S. Colla. 2014. Bumble Bees of North America. Princeton, NJ: Princeton University Press. 208 p. Wilson, J.S. and O.M. Carrill. 2016. The bees in your backyard; a guide to North America's bees. Princeton, NJ: Princeton University Press. 288 p.
Wilson, J.S. and O.M. Carrill. 2016. The bees in your backyard; a guide to North America's bees. Princeton, NJ: Princeton University Press. 288 p. Wojcik, V.A., G.W. Frankie, R.W. Thorp, and J.L. Hernandez. 2008. Seasonality in bees and their floral resource plants at a constructed urban bee habitat in Berkeley, California. Journal of the Kansas Entomological Society 81: 15-28.
Wojcik, V.A., G.W. Frankie, R.W. Thorp, and J.L. Hernandez. 2008. Seasonality in bees and their floral resource plants at a constructed urban bee habitat in Berkeley, California. Journal of the Kansas Entomological Society 81: 15-28.
- Additional ReferencesLegend:
 View Online Publication
View Online Publication
Do you know of a citation we're missing? Adhikari, S. 2018. Impacts of dryland farming systems on biodiversity, plant-insect interactions, and ecosystem services. Ph.D. Dissertation. Bozeman, MT: Montana State University. 207 p.
Adhikari, S. 2018. Impacts of dryland farming systems on biodiversity, plant-insect interactions, and ecosystem services. Ph.D. Dissertation. Bozeman, MT: Montana State University. 207 p. Adhikari, S., L.A. Burkle, K.M. O'Neill, C.M. Delphia, D.K. Weaver, and F.D. Menalled. 2019. Dryland organic farming partially offsets negative effects of highly simplified agricultural landscapes on forbs, bees, and bee-flower networks. Environmental Entomology 48(4): 826-835.
Adhikari, S., L.A. Burkle, K.M. O'Neill, C.M. Delphia, D.K. Weaver, and F.D. Menalled. 2019. Dryland organic farming partially offsets negative effects of highly simplified agricultural landscapes on forbs, bees, and bee-flower networks. Environmental Entomology 48(4): 826-835. Brutscher, L.M. and Flenniken, M.L. 2015. RNAi and antiviral defense in the Honey Bee. Journal of Immunology Research. 2015. 1-10. 10.1155/2015/941897.
Brutscher, L.M. and Flenniken, M.L. 2015. RNAi and antiviral defense in the Honey Bee. Journal of Immunology Research. 2015. 1-10. 10.1155/2015/941897. Brutscher, L.M., K.F. Daughenbaugh, M.L. Flenniken. 2015. Antiviral defense mechanisms in honey bees. Current Opinion in Insect Science 10:71-82. http://dx.doi.org/10.1016/j.cois.2015.04.016
Brutscher, L.M., K.F. Daughenbaugh, M.L. Flenniken. 2015. Antiviral defense mechanisms in honey bees. Current Opinion in Insect Science 10:71-82. http://dx.doi.org/10.1016/j.cois.2015.04.016 Brutscher, L.M., K.F. Daughenbaugh, M.L. Flenniken. 2017. Virus and dsRNA-triggered transcriptional responses reveal key components of honey bee antiviral defense. Scientific Reports 7:6448 https://doi.org/10.1038/s41598-017-06623-z
Brutscher, L.M., K.F. Daughenbaugh, M.L. Flenniken. 2017. Virus and dsRNA-triggered transcriptional responses reveal key components of honey bee antiviral defense. Scientific Reports 7:6448 https://doi.org/10.1038/s41598-017-06623-z Burkle L.A., M.P. Simanonok, J.S. Durney, J.A. Myers, and R.T. Belote. 2019. Wildfires influence abundance, diversity, and intraspecific and interspecific trait variation of native bees and flowering plants across burned and unburned landscapes. Frontiers in Ecology and Evolution 7(252):1-14.
Burkle L.A., M.P. Simanonok, J.S. Durney, J.A. Myers, and R.T. Belote. 2019. Wildfires influence abundance, diversity, and intraspecific and interspecific trait variation of native bees and flowering plants across burned and unburned landscapes. Frontiers in Ecology and Evolution 7(252):1-14. Daughenbaugh, K.F., M. Martin, L.M. Brutscher, I. Cavigli, E. Garcia, M. Lavin, and M.L. Flenniken. 2015. Honey bee infecting Lake Sinai viruses. Viruses 7, 3285-3309. https://doi.org/10.3390/v7062772
Daughenbaugh, K.F., M. Martin, L.M. Brutscher, I. Cavigli, E. Garcia, M. Lavin, and M.L. Flenniken. 2015. Honey bee infecting Lake Sinai viruses. Viruses 7, 3285-3309. https://doi.org/10.3390/v7062772 De Smet-Moens, H. 1982. The insect fauna of Canada Thistle Cirsium arvense (L.) Scop in southern Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 51 p.
De Smet-Moens, H. 1982. The insect fauna of Canada Thistle Cirsium arvense (L.) Scop in southern Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 51 p. Delphia, C.M., Griswold, T., Reese, E.G., O'Neill, K.M., and Burkle, L.A. 2019. Checklist of bees (Hymenoptera: Apoidea) from small, diversified vegetable farms in south-western Montana. Biodiversity Data Journal: e30062
Delphia, C.M., Griswold, T., Reese, E.G., O'Neill, K.M., and Burkle, L.A. 2019. Checklist of bees (Hymenoptera: Apoidea) from small, diversified vegetable farms in south-western Montana. Biodiversity Data Journal: e30062 Dolan, A.C. 2016. Insects associated with Montana's huckleberry (Ericaceae: Vaccinium globulare) plants and the bumble bees (Hymenoptera: Apidae) of Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 160 p.
Dolan, A.C. 2016. Insects associated with Montana's huckleberry (Ericaceae: Vaccinium globulare) plants and the bumble bees (Hymenoptera: Apidae) of Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 160 p. Doublet, V., Y. Poeschl, A. Gogol-Doring, C. Alaux, D. Annoscia, C. Aurori, S.M. Barribeau, O.C. Bedoya-Reina, M.J.F. Brown, J.C. Bull, M.L. Flenniken, D.A. Galbraith, E. Genersch, S. Gisder, I. Grosse, H.L. Holt, D. Hultmark, H.M. Lattorff, Y. Le Conte, F. Manfredini, D.P. McMahon, R.F.A. Moritz, F. Nazzi, E.L. Nino, K. Nowick, R.P. van Rij, R.J. Paxton, and C.M. Grozinger. 2017. Unity in defence: honeybee workers exhibit conserved molecular responses to diverse pathogens. BMC Genomics 18: 207. https://doi.org/10.1186/s12864-017-3597-6
Doublet, V., Y. Poeschl, A. Gogol-Doring, C. Alaux, D. Annoscia, C. Aurori, S.M. Barribeau, O.C. Bedoya-Reina, M.J.F. Brown, J.C. Bull, M.L. Flenniken, D.A. Galbraith, E. Genersch, S. Gisder, I. Grosse, H.L. Holt, D. Hultmark, H.M. Lattorff, Y. Le Conte, F. Manfredini, D.P. McMahon, R.F.A. Moritz, F. Nazzi, E.L. Nino, K. Nowick, R.P. van Rij, R.J. Paxton, and C.M. Grozinger. 2017. Unity in defence: honeybee workers exhibit conserved molecular responses to diverse pathogens. BMC Genomics 18: 207. https://doi.org/10.1186/s12864-017-3597-6 Flenniken, M.L. 2014. Honey Bee-Infecting Plant Virus with Implications on Honey Bee Colony Health. mBio 5(2):e00877-14. doi:10.1128/mBio.00877-14.
Flenniken, M.L. 2014. Honey Bee-Infecting Plant Virus with Implications on Honey Bee Colony Health. mBio 5(2):e00877-14. doi:10.1128/mBio.00877-14. Flenniken, M.L., R. Andino. 2013. Non-Specific dsRNA-Mediated Antiviral Response in the Honey Bee. PLOS ONE 8(10): e77263 doi:10.1371/ journal.pone.0077263
Flenniken, M.L., R. Andino. 2013. Non-Specific dsRNA-Mediated Antiviral Response in the Honey Bee. PLOS ONE 8(10): e77263 doi:10.1371/ journal.pone.0077263 Kearns, C.A. and J.D. Thomson. 2001. The Natural History of Bumble Bees. Boulder, CO. University Press of Colorado.
Kearns, C.A. and J.D. Thomson. 2001. The Natural History of Bumble Bees. Boulder, CO. University Press of Colorado. McMenamin, A.J., and M.L. Flenniken. 2018. Recently identified bee viruses and their impact on bee pollinators. Current Opinion in Insect Science 26:120-129. https://doi.org/10.1016/j.cois.2018.02.009
McMenamin, A.J., and M.L. Flenniken. 2018. Recently identified bee viruses and their impact on bee pollinators. Current Opinion in Insect Science 26:120-129. https://doi.org/10.1016/j.cois.2018.02.009 McMenamin, A.J., L.M. Brutscher, W. Glenny, and M.L. Flenniken. 2016. Abiotic and biotic factors affecting the replication and pathogenicity of bee viruses. Current Opinion in Insect Science 16:14-21.
McMenamin, A.J., L.M. Brutscher, W. Glenny, and M.L. Flenniken. 2016. Abiotic and biotic factors affecting the replication and pathogenicity of bee viruses. Current Opinion in Insect Science 16:14-21. Piccolomini, A.M., S.R. Whiten, M.L. Flenniken, K.M. O'Neill, and R.K.D. Peterson. 2018. Acute toxicity of permethrin, deltamethrin, and etofenprox to the alfalfa leafcutting bee. Journal of Economic Entomology 111(3):1001-1005 https://dx.doi.org/10.1093/jee/toy014
Piccolomini, A.M., S.R. Whiten, M.L. Flenniken, K.M. O'Neill, and R.K.D. Peterson. 2018. Acute toxicity of permethrin, deltamethrin, and etofenprox to the alfalfa leafcutting bee. Journal of Economic Entomology 111(3):1001-1005 https://dx.doi.org/10.1093/jee/toy014 Reese, E.G., L.A. Burkle, C.M. Delphia, and T. Griswold. 2018. A list of bees from three locations in the Northern Rockies Ecoregion (NRE) of western Montana. Biodiversity Data Journal 6: e27161.
Reese, E.G., L.A. Burkle, C.M. Delphia, and T. Griswold. 2018. A list of bees from three locations in the Northern Rockies Ecoregion (NRE) of western Montana. Biodiversity Data Journal 6: e27161. Runckel, C., J. DeRisi, and M.L. Flenniken. 2014. A draft genome of the honey bee trypanosomatid parasite Crithidia mellificae. PLoS ONE 9(4): e95057. doi:10.1371/journal.pone.0095057
Runckel, C., J. DeRisi, and M.L. Flenniken. 2014. A draft genome of the honey bee trypanosomatid parasite Crithidia mellificae. PLoS ONE 9(4): e95057. doi:10.1371/journal.pone.0095057 Runckel, C., M.L. Flenniken, J.C. Engel, J.G. Ruby, D. Ganem, R. Andino, and J.L. DeRisi. 2011. Temporal analysis of the honey bee microbiome reveals four novel viruses and seasonal prevalence of known viruses, Nosema, and Crithidia. PLoS ONE 6(6): e20656. doi:10.1371/journal.pone.0020656
Runckel, C., M.L. Flenniken, J.C. Engel, J.G. Ruby, D. Ganem, R. Andino, and J.L. DeRisi. 2011. Temporal analysis of the honey bee microbiome reveals four novel viruses and seasonal prevalence of known viruses, Nosema, and Crithidia. PLoS ONE 6(6): e20656. doi:10.1371/journal.pone.0020656 Sater, S. 2022. The insects of Sevenmile Creek, a pictorial guide to their diversity and ecology. Undergraduate Thesis. Helena, MT: Carroll College. 242 p.
Sater, S. 2022. The insects of Sevenmile Creek, a pictorial guide to their diversity and ecology. Undergraduate Thesis. Helena, MT: Carroll College. 242 p. Simanonok, M. 2018. Plant-pollinator network assembly after wildfire. Ph.D. Dissertation. Bozeman, MT: Montana State University. 123 p.
Simanonok, M. 2018. Plant-pollinator network assembly after wildfire. Ph.D. Dissertation. Bozeman, MT: Montana State University. 123 p. Simanonok, M.P. and L.A. Burkle. 2019. Nesting success of wood-cavity-nesting bees declines with increasing time since wildfire. Ecology and Evolution 9:12436-12445.
Simanonok, M.P. and L.A. Burkle. 2019. Nesting success of wood-cavity-nesting bees declines with increasing time since wildfire. Ecology and Evolution 9:12436-12445. Visscher, P.K. 1989. A quantitative study of worker reproduction in honey bee colonies. Behavioural Ecology and Sociobiology 25:247-254.
Visscher, P.K. 1989. A quantitative study of worker reproduction in honey bee colonies. Behavioural Ecology and Sociobiology 25:247-254. Visscher, P.K. and T.D. Seeley. 1989. Bee-lining as a research technique in ecological studies of honey bees. American Bee Journal. 129: 536-539.
Visscher, P.K. and T.D. Seeley. 1989. Bee-lining as a research technique in ecological studies of honey bees. American Bee Journal. 129: 536-539.
- Web Search Engines for Articles on "European Honey Bee"
- Additional Sources of Information Related to "Insects"





