View in other NatureServe Network Field Guides
NatureServe
Montana
Utah
Wyoming
Idaho
Wisconsin
British Columbia
South Carolina
Yukon
California
New York
Northern Redbelly Dace - Chrosomus eos
State Rank Reason (see State Rank above)
Species is facing significant threats from the invasion of northern pike and has undergone local extirpation and continues to decline.
General Description
The Northern Redbelly Dace is one of Montana's small native minnows found in small cool, prairie streams. During the spawning season, the males of this species become quite colorful with red flanks. Its maximum size is approximately 3 inches.
For a comprehensive review of the ecology, conservation status, threats, and management of this and other Montana fish species of concern, please see
Montana Chapter of the American Fisheries Society Species of Concern Status Reviews.Diagnostic Characteristics
The Northern Redbelly Dace is olive to dark brown above; the lower side and belly are yellow or silvery except on adult males during summer when the lower side is red. The side has two dark stripes with a light band between them; the upper stripe often breaks into spots toward the tail. The lower stripe is broader and extends from the snout to the base of the tail fin where it may end in a spot. These two lateral stripes are sometimes connected by a dark, oblique line or crossband. The eyes are large. The body is almost round in cross section. The front of the dorsal fin is behind the front of the pelvic fins. The lateral line is incomplete and not distinct and there are no barbels (Brown 1971, Holton and Johnson 1996).
Species Range
Montana Range
Range Descriptions
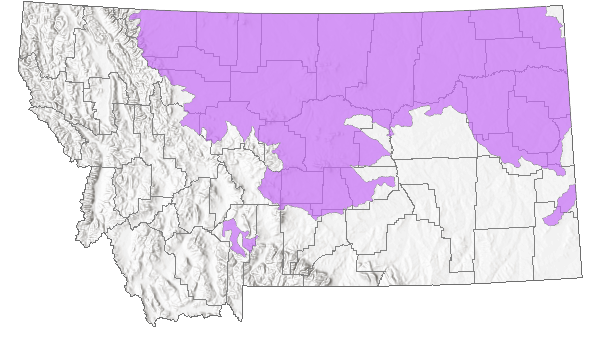
 Native
Native
Western Hemisphere Range
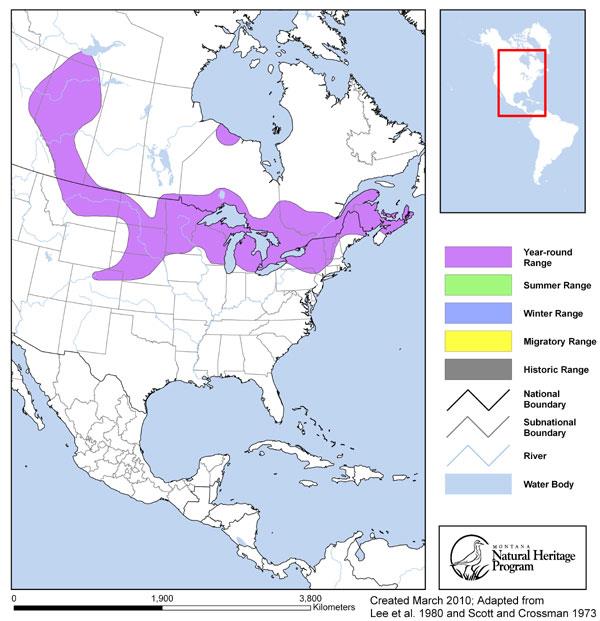
Range Comments
Northern Redbelly Dace range across Canada from British Columbia through the Northwest Territories to Nova Scotia, and from Montana to Maine, primarily in the Atlantic, Great Lakes, Hudson Bay, upper Mississippi, Missouri, and Peace-Mackenzie river drainages, with populations in the west south to northern Colorado.
Observations in Montana Natural Heritage Program Database
Number of Observations: 346
(Click on the following maps and charts to see full sized version)
Map Help and Descriptions
Relative Density
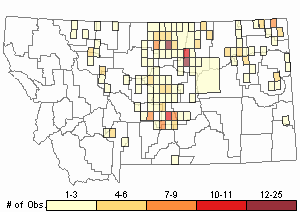
Recency
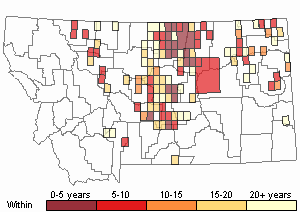
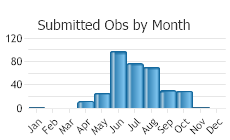
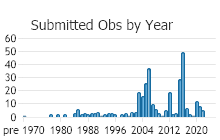
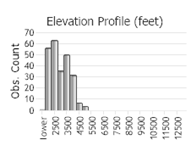 (Observations spanning multiple months or years are excluded from time charts)
(Observations spanning multiple months or years are excluded from time charts)
Migration
Not studied in Montana. In a 7.3 hectare lake in Quebec, Northern Redbelly Dace swam in shoals in the shallow littoral zone during the day and migrated to the deeper pelagic regions at sunset, where shoals broke up into single fish, then regrouped as individuals returned to the shallow littoral areas at sunrise; migrations began in late May (Naud and Magnan 1988). Patterns of movement in streams is apparently not well-documented, but probably includes moves of several hundred meters.
Habitat
Northern Redbelly Dace are found in clear, cool, slow-flowing creeks, ponds and lakes with aquatic vegetation, including filamentous algae, and sandy or gravelly bottoms interspersed with silt (Brown 1971). In Montana, this species is an indicator species of the Northern Glaciated Prairie Stream Ecological System and may occur in the intermittent prairie stream systems. In Stink Creek of northern South Dakota (Morey and Berry 2004), dace were found in a single unconstrained and low-gradient prairie channel with little surface flow at the time (early June). Pools were 26 to 84 cm (1.0 to 3.3 inches) deep in a wetted area about 1.3 m (4.3 ft) wide. Water temperature was 16.6 C, filamentous algae and rooted macrophytes were abundant. In western South Dakota, the range of this species appears to be limited to perrenial streams with slow, clear water and abundant macrophytes and algae (Morey and Berry 2004). In Colorado (Bestgen 1989), the presence of cool springs or cool tributary flow was a consistent feature of Northern Redbelly Dace habitats in prairie streams and marshes. Strong thermal stratification existed in some heavily vegetated ponds. In water < 0.3 m (0.9 ft) deep, surface temperature was 27 C when bottom temperature was 18 C; water < 22 C was present in all occupied habitats. Individuals were usually found in water 0.25 to 1.3 m (0.8 to 4.3 ft) deep and congregated near the shores of ponds >3.0 m (9.8 ft) deep. Substrate in all habitats ranged from black anoxic silt to sand.
Food Habits
Diet has not been studied in Montana. In a Quebec lake (Naud and Magnan 1988), 95% of the food items in 78 stomachs consisted of two genera of cladocerans (Daphnia and Holopedium). Contents (frequency of occurrence) in 40 stomachs from Michigan (Cochran et al. 1988) included green algae (100%), diatoms (68.4%), blue-green algae (10.5%), dinoflagellates (42.1%), zooplankton (42.1%: rotifers, copepods, Daphinia, Chaoborus, Bosmina), macroinvertebrates (5.3%: flies, caddisflies, beetles, mayflies) and miscellaneous (89.4%: sponge gemmules, pollen, macrophyte fragments).
Ecology
Not studied in Montana (but see Habitat comments). Age structure of collections from Colorado in August and September (Bestgen 1989), respectively, were Age Class 0 (13-35 mm standard length): 82% and 82.5%, Age Class 1 (33-46 mm): 12% and 15%, Age Class 2 (45-52 mm): 6% and 2.5%; some populations from Canada are reported to include fish living 5 to 8 years. In Colorado (Bestgen 1989), Northern Redbelly Dace were absent from sites when Green Sunfish and Largemouth Bass were present. Risk of predation appears to be an important agent for Northern Redbelly Dace in Canadian Shield lakes. Northern Redbelly Dace are also subject to predation by Brook Trout in Canada, and appear to be sensitive to chemical alarm cues, by which they are able estimate the risk of predation (Dupuch et al. 2004). In Quebec (Dupuch et al. 2009), results of a study of 13 lakes showed that 1) mean abundance of dace in the littoral zone was positively related with the relative risk of predation in the pelagic zone, 2) dace preferentially used structured over open water habitats in the littoral zone, and 3) this preference was not related to the relative predation risk in the littoral zone, but decreased as the relative risk of predation increased in the pelagic zone. It was concluded that, at the lake level, dace appear to enter the littoral to avoid pelagic predators; at the littoral zone level, the results indicate flexibility in antipredator tactics (shelter versus shoaling). Movements to pelagic habitat at night for feeding and returning to littoral habitats during day may also be a way to reduce predation risk to themselves while increasing encounters with preferred prey (Naud and Magnan 1988).
Reproductive Characteristics
Not well-studied in Montana. The Northern Redbelly Dace is sexually mature at 1 year and spawns May through August. Incubation is 8 to 10 days at 70 to 80 degrees F (Brown 1971). In Colorado (Bestgen 1989), spawning appears to occur from late June to early August, although spawning is not necessarily synchronous in geographically close habitats, probably due to differences in water temperatures between spawning sites. In Alberta (Das and Nelson 1990), spawning extends from mid-June to late July at surface water temperatures of 13.0 to 18.0 C. Female gonads are ripe from early June to late July, fully ripe from mid-June to late July, and spent from mid-June to mid-August. The number of eggs carried by 50 female dace 39.4-54.5 mm standard length was 146-1030 (mean = 654). Fully ripe eggs are 0.91 to 1.24 mm diameter and golden yellow. Eggs are scattered among aquatic vegetation over gravelly substrates and abandoned. A protracted spawning period in Ontario (and elsewhere), the patterns of fecundity to size relationships (lack of a relationship between female length and number of eggs carried), and egg size distributions with no apparent age (size) differentiation, suggest that Northern Redbelly Dace is a fractional (multiple, serial, or batch) spawner in at least some parts of its range (Powles et al. 1992).
In some locations in the northern U.S. and Canada, the Northern Redbelly Dace hybridizes with its close relative, the Finescale Dace. The resultant hybrids are very unusual in that they are all females and produce offspring that are likewise all female. Eggs from the hybrids are "fertilized" by the sperm of Northern Redbelly Dace. It appears that this "fertilization" is necessary for egg development to begin, but curiously none of the genetic traits of the male are incorporated into the fertilized embryo. This type of reproduction is known as gynogenesis and is found in only a few fish and amphibians. Here in Montana, we have the Northern Redbelly Dace and the hybrids, but no Finescale Dace.
Management
No management activities specific to Northern Redbelly Dace are currently occuring in Montana. Surveys to assess population status and life histroy throughout the state would be useful. Most assessments have been to determine status and genetic structure of hybrid Northern Redbelly X Finescale Dace. Presence in livestock watering ponds in Colorado (Bestgen 1989) indicate an ability to coexist with livestock under certain conditions.
Stewardship Responsibility
Threats or Limiting Factors
As with many small native stream fishes, Northern Redbelly Dace could be adversely effected by stream channelization, reductions to discharge, changes in water quality and temperature, and introductions of non-native predatory fishes. Northern pike introduction across the glaciated prairie streams of northern Montana have significantly affected the small native minnow communities (Stagliano 2008).
References
- Literature Cited AboveLegend:
 View Online Publication
View Online Publication Bestgen, K. R. 1989. Distribution and notes on the biology of Phoxinus eos (Cyprinidae) in Colorado. Southwestern Naturalist 34(2):225-231.
Bestgen, K. R. 1989. Distribution and notes on the biology of Phoxinus eos (Cyprinidae) in Colorado. Southwestern Naturalist 34(2):225-231. Brown, C.J.D. 1971. Fishes of Montana. Bozeman, MT: Big Sky Books/Montana State University. 207 p.
Brown, C.J.D. 1971. Fishes of Montana. Bozeman, MT: Big Sky Books/Montana State University. 207 p. Cochran, P. A., D. M. Lodge, J. R. Hodgson, and P. G. Knapik. 1988. Diets of syntopic finescale dace, Phoxinus neogaeus, and northern redbelly dace, Phoxinus eos: a reflection of trophic morphology. Environmental Biology of Fishes 22 (3): 235-240.
Cochran, P. A., D. M. Lodge, J. R. Hodgson, and P. G. Knapik. 1988. Diets of syntopic finescale dace, Phoxinus neogaeus, and northern redbelly dace, Phoxinus eos: a reflection of trophic morphology. Environmental Biology of Fishes 22 (3): 235-240. Das, M.K. and J.S. Nelson. 1990. Spawning time and fecundity of northern redbelly dace (Phoxinus neogaeus), finescale dace (Phoxinus neogaeus), and their hybrids in Upper Pierre Grey Lake, Alberta. Canadian Field Naturalist 104(3):409-413.
Das, M.K. and J.S. Nelson. 1990. Spawning time and fecundity of northern redbelly dace (Phoxinus neogaeus), finescale dace (Phoxinus neogaeus), and their hybrids in Upper Pierre Grey Lake, Alberta. Canadian Field Naturalist 104(3):409-413. Dupuch A., P. Magnan, A. Bertolo, L.M. Dill, and M. Proulx. 2009. Does predation risk influence habitat use by northern redbelly dace Phoxinus eos at different spatial scales? Journal of Fish Biology 74 (7): 1371-82.
Dupuch A., P. Magnan, A. Bertolo, L.M. Dill, and M. Proulx. 2009. Does predation risk influence habitat use by northern redbelly dace Phoxinus eos at different spatial scales? Journal of Fish Biology 74 (7): 1371-82. Dupuch, A., P. Magnan, and L. M. Dill. 2004. Sensitivity of northern redbelly dace, Phoxinus eos, to chemical alarm cues. Canadian Journal of Zoology 82(3): 407-415.
Dupuch, A., P. Magnan, and L. M. Dill. 2004. Sensitivity of northern redbelly dace, Phoxinus eos, to chemical alarm cues. Canadian Journal of Zoology 82(3): 407-415. Holton, G. D. and H. E. Johnson. 1996. A field guide to Montana fishes. Second Edition. Montana Department of Fish, Wildlife and Parks, Helena. 104 pp.
Holton, G. D. and H. E. Johnson. 1996. A field guide to Montana fishes. Second Edition. Montana Department of Fish, Wildlife and Parks, Helena. 104 pp. Lee, D.S., C.R. Gilbert, C.H. Hocutt, R.E. Jenkins, D. E. McAllister, J. R. Stauffer, Jr. 1980. Atlas of North American freshwater fishes. North Carolina State Musuem of Natural History. 867 p.
Lee, D.S., C.R. Gilbert, C.H. Hocutt, R.E. Jenkins, D. E. McAllister, J. R. Stauffer, Jr. 1980. Atlas of North American freshwater fishes. North Carolina State Musuem of Natural History. 867 p. Morey, N. M. and C. R. Berry. 2004. New distributional record of the northern redbelly dace in the northern great plains. The Prairie Naturalist 36: 257-260.
Morey, N. M. and C. R. Berry. 2004. New distributional record of the northern redbelly dace in the northern great plains. The Prairie Naturalist 36: 257-260. Naud, M. and P. Magnan. 1988. Diel onshore–offshore migrations in northern redbelly dace, Phoxinus eos (cope), in relation to prey distribution in a small oligotrophic lake. Canadian Journal of Zoology 66 (6): 1249-1253.
Naud, M. and P. Magnan. 1988. Diel onshore–offshore migrations in northern redbelly dace, Phoxinus eos (cope), in relation to prey distribution in a small oligotrophic lake. Canadian Journal of Zoology 66 (6): 1249-1253. Powles, P. M., S. Finucan, M. Van Haaften, and R. A. Curry. 1992. Preliminary evidence for fractional spawning by the northern redbelly dace, Phoxinus eos. Canadian Field Naturalist 106(2): 237.
Powles, P. M., S. Finucan, M. Van Haaften, and R. A. Curry. 1992. Preliminary evidence for fractional spawning by the northern redbelly dace, Phoxinus eos. Canadian Field Naturalist 106(2): 237. Scott, W.B. and E.J. Crossman. 1973. Rainbow trout, Kamloops trout, Steelhead trout Salmo gairdneri Richardson. pp. 184-191. In: Freshwater fishes of Canada. Ottawa, Canada: Fisheries Research Board of Canada, Bulletin 184. 966 p.
Scott, W.B. and E.J. Crossman. 1973. Rainbow trout, Kamloops trout, Steelhead trout Salmo gairdneri Richardson. pp. 184-191. In: Freshwater fishes of Canada. Ottawa, Canada: Fisheries Research Board of Canada, Bulletin 184. 966 p.
- Additional ReferencesLegend:
 View Online Publication
View Online Publication
Do you know of a citation we're missing? Barfoot, C.A. 1993. Longitudinal distribution of fishes and habitat in Little Beaver Creek, Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 66 p.
Barfoot, C.A. 1993. Longitudinal distribution of fishes and habitat in Little Beaver Creek, Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 66 p. Dieterman, D.J., M.P. Ruggles, M.L. Wildhaber, and D.L. Galat (eds). 1996. Population structure and habitat use of benthic fishes along the Missouri and Lower Yellowstone Rivers. 1996 Annual report of Missouri River Benthic Fish Study PD-95-5832 to U.S. Army Corps of Engineers and U.S. Bureau of Reclamation. 238 p.
Dieterman, D.J., M.P. Ruggles, M.L. Wildhaber, and D.L. Galat (eds). 1996. Population structure and habitat use of benthic fishes along the Missouri and Lower Yellowstone Rivers. 1996 Annual report of Missouri River Benthic Fish Study PD-95-5832 to U.S. Army Corps of Engineers and U.S. Bureau of Reclamation. 238 p. Endicott, C.L. 1996. Responses of riparian and stream ecosystems to varying timing and intensity of livestock grazing in central Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 115 p.
Endicott, C.L. 1996. Responses of riparian and stream ecosystems to varying timing and intensity of livestock grazing in central Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 115 p. Hendricks, P., S. Lenard, D.M. Stagliano, and B.A. Maxell. 2013. Baseline nongame wildlife surveys on the Fort Peck Indian Reservation. Report to the Assiniboine and Sioux Tribes of the Fort Peck Indian Reservation. Montana Natural Heritage Program, Helena, MT. 83 p.
Hendricks, P., S. Lenard, D.M. Stagliano, and B.A. Maxell. 2013. Baseline nongame wildlife surveys on the Fort Peck Indian Reservation. Report to the Assiniboine and Sioux Tribes of the Fort Peck Indian Reservation. Montana Natural Heritage Program, Helena, MT. 83 p. Joslin, Gayle, and Heidi B. Youmans. 1999. Effects of recreation on Rocky Mountain wildlife: a review for Montana. [Montana]: Montana Chapter of the Wildlife Society.
Joslin, Gayle, and Heidi B. Youmans. 1999. Effects of recreation on Rocky Mountain wildlife: a review for Montana. [Montana]: Montana Chapter of the Wildlife Society. Montana Chapter of the American Fisheries Society species status accounts.
Montana Chapter of the American Fisheries Society species status accounts. Mullins, M.S. 1991. Biology and predator use of cisco (Coregonus artedi) in Fort Peck Reservoir, Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 68 p.
Mullins, M.S. 1991. Biology and predator use of cisco (Coregonus artedi) in Fort Peck Reservoir, Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 68 p. Rosenthal, L.R. 2007. Evaluation of distribution and fish passage in relation to road culverts in two eastern Montana prairie streams. M.Sc. Thesis. Bozeman, MT: Montana State University. 78 p.
Rosenthal, L.R. 2007. Evaluation of distribution and fish passage in relation to road culverts in two eastern Montana prairie streams. M.Sc. Thesis. Bozeman, MT: Montana State University. 78 p. Stash, S.W. 2001. Distribution, relative abundance, and habitat associations of Milk River fishes related to irrigation diversion dams. M.Sc. Thesis. Bozeman, MT: Montana State University. 82 p.
Stash, S.W. 2001. Distribution, relative abundance, and habitat associations of Milk River fishes related to irrigation diversion dams. M.Sc. Thesis. Bozeman, MT: Montana State University. 82 p. Stringer, A. and N. Clancy. 2020. Status of Northern Redbelly Dace (Chrosomus eos) in Montana. Montana Chapter, American Fisheries Society.
Stringer, A. and N. Clancy. 2020. Status of Northern Redbelly Dace (Chrosomus eos) in Montana. Montana Chapter, American Fisheries Society. Stringer, A.L. 2018. Status of Northern Pearl Dace and chrosomid dace in prairie streams of Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 150 p.
Stringer, A.L. 2018. Status of Northern Pearl Dace and chrosomid dace in prairie streams of Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 150 p. Wuellner, M.R. 2007. Influence of reach and watershed characteristics on fish distributions in small streams of eastern Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 80 p.
Wuellner, M.R. 2007. Influence of reach and watershed characteristics on fish distributions in small streams of eastern Montana. M.Sc. Thesis. Bozeman, MT: Montana State University. 80 p.
- Web Search Engines for Articles on "Northern Redbelly Dace"
- Additional Sources of Information Related to "Fish"





